Olfactory navigation in arthropods
- PMID: 36658447
- PMCID: PMC10354148
- DOI: 10.1007/s00359-022-01611-9
Olfactory navigation in arthropods
Abstract
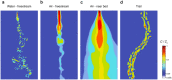
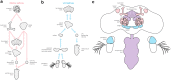
Using odors to find food and mates is one of the most ancient and highly conserved behaviors. Arthropods from flies to moths to crabs use broadly similar strategies to navigate toward odor sources-such as integrating flow information with odor information, comparing odor concentration across sensors, and integrating odor information over time. Because arthropods share many homologous brain structures-antennal lobes for processing olfactory information, mechanosensors for processing flow, mushroom bodies (or hemi-ellipsoid bodies) for associative learning, and central complexes for navigation, it is likely that these closely related behaviors are mediated by conserved neural circuits. However, differences in the types of odors they seek, the physics of odor dispersal, and the physics of locomotion in water, air, and on substrates mean that these circuits must have adapted to generate a wide diversity of odor-seeking behaviors. In this review, we discuss common strategies and specializations observed in olfactory navigation behavior across arthropods, and review our current knowledge about the neural circuits subserving this behavior. We propose that a comparative study of arthropod nervous systems may provide insight into how a set of basic circuit structures has diversified to generate behavior adapted to different environments.
Keywords: Arthropod; Insect; Navigation; Neural circuits; Olfaction.
© 2023. The Author(s).
Conflict of interest statement
The authors declare that they have no conflict of interest
Figures



References
-
- Ache BW, Derby CD. Functional organization of olfaction in crustaceans. Trends Neurosci. 1985;8:356–360. doi: 10.1016/0166-2236(85)90122-5. - DOI
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources

