Non-Ewald methods for evaluating the electrostatic interactions of charge systems: similarity and difference
- PMID: 36659982
- PMCID: PMC9842848
- DOI: 10.1007/s12551-022-01029-2
Non-Ewald methods for evaluating the electrostatic interactions of charge systems: similarity and difference
Abstract
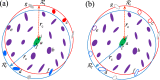
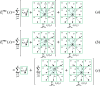
In molecular simulations, it is essential to properly calculate the electrostatic interactions of particles in the physical system of interest. Here we consider a method called the non-Ewald method, which does not rely on the standard Ewald method with periodic boundary conditions, but instead relies on the cutoff-based techniques. We focus on the physicochemical and mathematical conceptual aspects of the method in order to gain a deeper understanding of the simulation methodology. In particular, we take into account the reaction field (RF) method, the isotropic periodic sum (IPS) method, and the zero-multipole summation method (ZMM). These cutoff-based methods are based on different physical ideas and are completely distinguishable in their underlying concepts. The RF and IPS methods are "additive" methods that incorporate information outside the cutoff region, via dielectric medium and isotropic boundary condition, respectively. In contrast, the ZMM is a "subtraction" method that tries to remove the artificial effects, generated near the boundary, from the cutoff sphere. Nonetheless, we find physical and/or mathematical similarities between these methods. In particular, the modified RF method can be derived by the principle of neutralization utilized in the ZMM, and we also found a direct relationship between IPS and ZMM.
Keywords: Coulombic energy; Cutoff-based method; Electrostatic interaction; Electrostatic neutrality; Molecular dynamics; Non-Ewald method; Reaction field method.
© International Union for Pure and Applied Biophysics (IUPAB) and Springer-Verlag GmbH Germany, part of Springer Nature 2023, Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
Conflict of interest statement
Conflict of interestThe authors declare no competing interests.
Figures




References
-
- Abeyratne S, Gee A, Erdelyi B. An adaptive fast multipole method in Cartesian basis, enabled by algorithmic differentiation. Commun Nonlinear Sci Numer Simul. 2019;72:294–317. doi: 10.1016/j.cnsns.2019.01.001. - DOI
-
- Allen MP, Tildesley DJ. Computer simulation of liquids. Oxford: Clarendon; 1987.
-
- Allen MP, Tildesley DJ. Computer simulation of liquids. Second ed. Oxford: Oxford University Press; 2017.
-
- Alouges F, Aussal M. The sparse cardinal sine decomposition and its application for fast numerical convolution. Numerical Algorithms. 2015;70:427–448. doi: 10.1007/s11075-014-9953-6. - DOI
-
- Alper HE, Levy RM. Computer simulations of the dielectric properties of water: studies of the simple point charge and transferrable intermolecular potential models. J Chem Phys. 1989;91:1242–1251. doi: 10.1063/1.457198. - DOI
Publication types
LinkOut - more resources
Full Text Sources

