Presence of Spodoptera frugiperda Multiple Nucleopolyhedrovirus (SfMNPV) Occlusion Bodies in Maize Field Soils of Mesoamerica
- PMID: 36662012
- PMCID: PMC9864064
- DOI: 10.3390/insects14010080
Presence of Spodoptera frugiperda Multiple Nucleopolyhedrovirus (SfMNPV) Occlusion Bodies in Maize Field Soils of Mesoamerica
Abstract
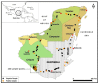
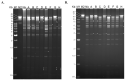
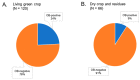
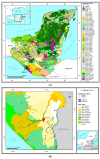
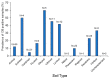
The occlusion bodies (OBs) of lepidopteran nucleopolyhedroviruses can persist in soil for extended periods before being transported back on to the foliage for transmission to the host insect. A sensitive insect bioassay technique was used to detect OBs of Spodoptera frugiperda multiple nucleopolyhedrovirus (SfMNPV) in 186 soil samples collected from maize fields in the southern Mexican states of Chiapas, Tabasco, Campeche, Yucatán, and Quintana Roo, as well Belize and Guatemala. Overall, 35 (18.8%) samples proved positive for SfMNPV OBs. The frequency of OB-positive samples varied significantly among Mexican states and countries (p < 0.05). Between 1.7 and 4.4% of S. frugiperda larvae that consumed OB-positive samples died from polyhedrosis disease. Restriction endonuclease analysis using PstI and HindIII confirmed that the soil-derived isolates were strains of SfMNPV and that genetic diversity was evident among the isolates. The prevalence of OB-positive soil samples did not differ with altitude or extension (area) of the maize field, but it was significantly higher in fields with the presence of living maize plants compared to those containing dead plants or crop residues (p < 0.05). Georeferenced soil samples were used to identify soil types on digitized soil maps. Lithosol and Luvisol soils had a higher than average prevalence of OB-positive samples (42−45% positive) (p = 0.006), as did Andosol, Gleysol, and Vertisol soils (33−60% OB-positive), although the sample sizes were small (<5 samples) for the latter three soils. In contrast, Cambisol soils had a lower than average prevalence of OB-positive samples (5% positive). Bioassays on Acrisol, Fluvisol, Phaeozem, and Rendzina soils resulted in intermediate levels of OB-positive samples. We conclude that certain soil types may favor OB persistence and virus-mediated biological pest control. The soil is also likely to provide a valuable source of genetic diversity for the design of virus-based insecticides against this pest.
Keywords: Alphabaculovirus; Baculoviridae; fall armyworm; genetic diversity; insect bioassay; soil type; virus persistence.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
Molecular and Biological Characterization of Spodoptera frugiperda Multiple Nucleopolyhedrovirus Field Isolate and Genotypes from China.Insects. 2020 Nov 10;11(11):777. doi: 10.3390/insects11110777. Insects. 2020. PMID: 33182689 Free PMC article.
-
Differential insecticidal properties of Spodoptera frugiperda multiple nucleopolyhedrovirus isolates against corn-strain and rice-strain fall armyworm, and genomic analysis of three isolates.J Invertebr Pathol. 2021 Jul;183:107561. doi: 10.1016/j.jip.2021.107561. Epub 2021 Feb 25. J Invertebr Pathol. 2021. PMID: 33639152
-
Analysis of a naturally-occurring deletion mutant of Spodoptera frugiperda multiple nucleopolyhedrovirus reveals sf58 as a new per os infectivity factor of lepidopteran-infecting baculoviruses.J Invertebr Pathol. 2012 Jan;109(1):117-26. doi: 10.1016/j.jip.2011.10.010. Epub 2011 Oct 21. J Invertebr Pathol. 2012. PMID: 22041202
-
Genetic variants in Argentinean isolates of Spodoptera frugiperda Multiple Nucleopolyhedrovirus.Virus Genes. 2020 Jun;56(3):401-405. doi: 10.1007/s11262-020-01741-9. Epub 2020 Feb 6. Virus Genes. 2020. PMID: 32030574
-
Pathogenic Assessment of SfMNPV-Based Biopesticide on Spodoptera frugiperda (Lepidoptera: Noctuidae) Developing on Transgenic Soybean Expressing Cry1Ac Insecticidal Protein.J Econ Entomol. 2021 Dec 6;114(6):2264-2270. doi: 10.1093/jee/toab170. J Econ Entomol. 2021. PMID: 34487171
Cited by
-
A qPCR Assay for the Quantification of Selected Genotypic Variants of Spodoptera frugiperda Multiple Nucleopolyhedrovirus (Baculoviridae).Viruses. 2024 May 30;16(6):881. doi: 10.3390/v16060881. Viruses. 2024. PMID: 38932173 Free PMC article.
-
Baculovirus Genetic Diversity and Population Structure.Viruses. 2025 Jan 22;17(2):142. doi: 10.3390/v17020142. Viruses. 2025. PMID: 40006898 Free PMC article. Review.
-
Fecal Transmission of Spodoptera frugiperda Multiple Nucleopolyhedrovirus (SfMNPV; Baculoviridae).Viruses. 2025 Feb 21;17(3):298. doi: 10.3390/v17030298. Viruses. 2025. PMID: 40143229 Free PMC article.
-
Morphological, Biological, and Molecular Characterization of Type I Granuloviruses of Spodoptera frugiperda.Neotrop Entomol. 2024 Aug;53(4):917-928. doi: 10.1007/s13744-024-01172-3. Epub 2024 Jun 28. Neotrop Entomol. 2024. PMID: 38940947
References
-
- D’Amico V., Elkinton J.S. Rainfall effects on transmission of gypsy moth (Lepidoptera: Lymantriidae) nuclear polyhedrosis virus. Environ. Entomol. 1995;24:1144–1149. doi: 10.1093/ee/24.5.1144. - DOI
-
- Fuxa J.R., Richter A.R. Effect of agricultural operations and precipitation on vertical distribution of a nuclear polyhedrosis virus in soil. Biol. Control. 1996;6:324–329. doi: 10.1006/bcon.1996.0041. - DOI
-
- Williams T. Viruses. In: Hajek A.E., Shapiro-Ilan D.I., editors. Ecology of Invertebrate Diseases. Wiley; Chichester, UK: 2018. pp. 215–285.
-
- Fuxa J.R. Ecology of insect nucleopolyhedroviruses. Agric. Ecosyst. Environ. 2004;103:27–43. doi: 10.1016/j.agee.2003.10.013. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

