Inhibition of Voltage-Gated Na+ Currents Exerted by KB-R7943 (2-[2-[4-(4-nitrobenzyloxy)phenyl]ethyl]isothiourea), an Inhibitor of Na+-Ca2+ Exchanging Process
- PMID: 36675319
- PMCID: PMC9864174
- DOI: 10.3390/ijms24021805
Inhibition of Voltage-Gated Na+ Currents Exerted by KB-R7943 (2-[2-[4-(4-nitrobenzyloxy)phenyl]ethyl]isothiourea), an Inhibitor of Na+-Ca2+ Exchanging Process
Abstract
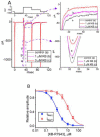
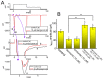
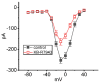
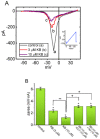
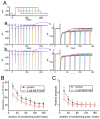
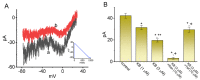
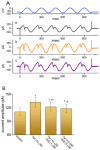
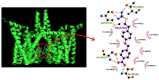
KB-R7943, an isothiourea derivative, has been recognized as an inhibitor in the reverse mode of the Na+-Ca2+ exchanging process. This compound was demonstrated to prevent intracellular Na+-dependent Ca2+ uptake in intact cells; however, it is much less effective at preventing extracellular Na+-dependent Ca2+ efflux. Therefore, whether or how this compound may produce any perturbations on other types of ionic currents, particularly on voltage-gated Na+ current (INa), needs to be further studied. In this study, the whole-cell current recordings demonstrated that upon abrupt depolarization in pituitary GH3 cells, the exposure to KB-R7943 concentration-dependently depressed the transient (INa(T)) or late component (INa(L)) of INa with an IC50 value of 11 or 0.9 μM, respectively. Likewise, the dissociation constant for the KB-R7943-mediated block of INa on the basis of a minimum reaction scheme was estimated to be 0.97 μM. The presence of benzamil or amiloride could suppress the INa(L) magnitude. The instantaneous window Na+ current (INa(W)) activated by abrupt ascending ramp voltage (Vramp) was suppressed by adding KB-R7943; however, subsequent addition of deltamethrin or tefluthrin (Tef) effectively reversed KB-R7943-inhibted INa(W). With prolonged duration of depolarizing pulses, the INa(L) amplitude became exponentially decreased; moreover, KB-R7943 diminished INa(L) magnitude. The resurgent Na+ current (INa(R)) evoked by a repolarizing Vramp was also suppressed by adding this compound; moreover, subsequent addition of ranolazine or Tef further diminished or reversed, respectively, its reduction in INa(R) magnitude. The persistent Na+ current (INa(P)) activated by sinusoidal voltage waveform became enhanced by Tef; however, subsequent application of KB-R7943 counteracted Tef-stimulated INa(P). The docking prediction reflected that there seem to be molecular interactions of this molecule with the hNaV1.2 or hNaV1.7 channels. Collectively, this study highlights evidence showing that KB-R7943 has the propensity to perturb the magnitude and gating kinetics of INa (e.g., INa(T), INa(L), INa(W), INa(R), and INa(P)) and that the NaV channels appear to be important targets for the in vivo actions of KB-R7943 or other relevant compounds.
Keywords: KB-R7943; Na+-Ca2+ exchange; current kinetics; late Na+ current; persistent Na+ current; resurgent Na+ current; transient Na+ current; voltage-gated Na+ current; window Na+ current.
Conflict of interest statement
No conflicts of interest, financial or otherwise, are declared by the author(s). The content and writing of this paper are solely the responsibility of the authors.
Figures
Similar articles
-
Concerted suppressive effects of carisbamate, an anti-epileptic alkyl-carbamate drug, on voltage-gated Na+ and hyperpolarization-activated cation currents.Front Cell Neurosci. 2023 May 24;17:1159067. doi: 10.3389/fncel.2023.1159067. eCollection 2023. Front Cell Neurosci. 2023. PMID: 37293624 Free PMC article.
-
Characterization in Inhibitory Effectiveness of Carbamazepine in Voltage-Gated Na+ and Erg-Mediated K+ Currents in a Mouse Neural Crest-Derived (Neuro-2a) Cell Line.Int J Mol Sci. 2022 Jul 17;23(14):7892. doi: 10.3390/ijms23147892. Int J Mol Sci. 2022. PMID: 35887240 Free PMC article.
-
The Evidence for Effective Inhibition of INa Produced by Mirogabalin ((1R,5S,6S)-6-(aminomethyl)-3-ethyl-bicyclo [3.2.0] hept-3-ene-6-acetic acid), a Known Blocker of CaV Channels.Int J Mol Sci. 2022 Mar 31;23(7):3845. doi: 10.3390/ijms23073845. Int J Mol Sci. 2022. PMID: 35409204 Free PMC article.
-
Effective Perturbations by Small-Molecule Modulators on Voltage-Dependent Hysteresis of Transmembrane Ionic Currents.Int J Mol Sci. 2022 Aug 21;23(16):9453. doi: 10.3390/ijms23169453. Int J Mol Sci. 2022. PMID: 36012718 Free PMC article. Review.
-
Late sodium current and calcium homeostasis in arrhythmogenesis.Channels (Austin). 2021 Dec;15(1):1-19. doi: 10.1080/19336950.2020.1854986. Channels (Austin). 2021. PMID: 33258400 Free PMC article. Review.
Cited by
-
Towards Novel Potential Molecular Targets for Antidepressant and Antipsychotic Pharmacotherapies.Int J Mol Sci. 2023 May 30;24(11):9482. doi: 10.3390/ijms24119482. Int J Mol Sci. 2023. PMID: 37298431 Free PMC article. Review.
-
Ion Channels as a Potential Target in Pharmaceutical Designs.Int J Mol Sci. 2023 Mar 30;24(7):6484. doi: 10.3390/ijms24076484. Int J Mol Sci. 2023. PMID: 37047455 Free PMC article.
-
Recent Advances in Ionic Mechanisms in Pituitary Cells: Implications for Electrophysiological and Electropharmacological Research.J Clin Med. 2025 Apr 30;14(9):3117. doi: 10.3390/jcm14093117. J Clin Med. 2025. PMID: 40364147 Free PMC article. Review.
-
Characterization of Stimulatory Action on Voltage-Gated Na+ Currents Caused by Omecamtiv Mecarbil, Known to Be a Myosin Activator.Biomedicines. 2023 May 3;11(5):1351. doi: 10.3390/biomedicines11051351. Biomedicines. 2023. PMID: 37239022 Free PMC article.
-
The effects of voltage dependence and ion-binding reaction rates on a thermodynamically constrained mathematical model of the Na/Ca exchanger.Sci Rep. 2025 Jul 1;15(1):21122. doi: 10.1038/s41598-025-07881-y. Sci Rep. 2025. PMID: 40596613 Free PMC article.
References
-
- Sung R.J., Wu Y.H., Lai N.H., Teng C.H., Luo C.H., Tien H.C., Lo C.P., Wu S.N. Beta-adrenergic modulation of arrhythmogenesis and identification of targeted sites of antiarrhythmic therapy in Timothy (LQT8) syndrome: A theoretical study. Am. J. Physiol. Heart Circ. Physiol. 2010;298:H33–H44. doi: 10.1152/ajpheart.00232.2009. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

