p-Sulfonato-Calix[4]arene Micelles Stabilize a Povidone Iodine Solution: Supramolecular Interactions, Iodine Retention, and Bactericidal Activity
- PMID: 36678039
- PMCID: PMC9865561
- DOI: 10.3390/nano13020286
p-Sulfonato-Calix[4]arene Micelles Stabilize a Povidone Iodine Solution: Supramolecular Interactions, Iodine Retention, and Bactericidal Activity
Abstract
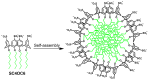
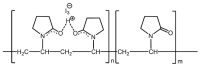
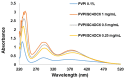
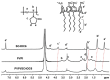
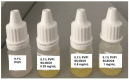
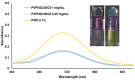
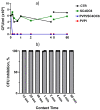
Povidone iodine (PVPI) is an antiseptic widely used against a broad spectrum of pathogens. However, undesired side-effects are still associated with PVPI treatment due to the irritant effect of iodine. Reducing the concentration of a PVPI formulation could provide safer and more friendly formulations, for routine use and applications in very delicate organs such as the eye. However, managing the storage of a low-concentration solution of PVPI is challenging due to the high iodine volatility. In this study, we demonstrated that an amphiphilic p-sulfonato-calix[4]arene derivative forming micelles (SC4OC6) improves the stability of a 0.1% PVPI aqueous buffered solution. UV-vis and NMR spectra as well as dynamic and electrophoretic light scattering measurements showed that SC4OC6 establishes non-covalent supramolecular interactions with PVPI, resulting in the formation of nanoaggregates with a negatively charged surface. Isothermal titration calorimetry provided the aggregation parameters and evidenced that the formation of the supramolecular assembly is an enthalpically favored process. The interaction of SC4OC6 with PVPI enhances the iodine retention and stability of the solution without affecting the rapid and effective bactericidal activity of PVPI, as demonstrated by a time-killing assay with Staphylococcus epidermidis.
Keywords: antibacterial activity; micellar aggregate; non-covalent interactions; povidone-iodine; sulfonato-calix[4]arene.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




References
-
- Gottardi W. Iodine and iodine compounds. In: Block S.S., editor. Disinfection, Sterilization and Preservation. 4th ed. Lea & Febiger; Philadelphia, PA, USA: 1991. pp. 152–166.
Grants and funding
LinkOut - more resources
Full Text Sources

