Recent Developments in DNA-Nanotechnology-Powered Biosensors for Zika/Dengue Virus Molecular Diagnostics
- PMID: 36678114
- PMCID: PMC9864780
- DOI: 10.3390/nano13020361
Recent Developments in DNA-Nanotechnology-Powered Biosensors for Zika/Dengue Virus Molecular Diagnostics
Abstract
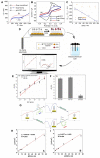
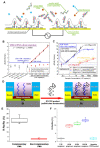
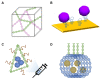
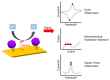
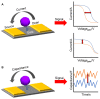
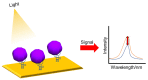
Zika virus (ZIKV) and dengue virus (DENV) are highly contagious and lethal mosquito-borne viruses. Global warming is steadily increasing the probability of ZIKV and DENV infection, and accurate diagnosis is required to control viral infections worldwide. Recently, research on biosensors for the accurate diagnosis of ZIKV and DENV has been actively conducted. Moreover, biosensor research using DNA nanotechnology is also increasing, and has many advantages compared to the existing diagnostic methods, such as polymerase chain reaction (PCR) and enzyme-linked immunosorbent assay (ELISA). As a bioreceptor, DNA can easily introduce a functional group at the 5' or 3' end, and can also be used as a folded structure, such as a DNA aptamer and DNAzyme. Instead of using ZIKV and DENV antibodies, a bioreceptor that specifically binds to viral proteins or nucleic acids has been fabricated and introduced using DNA nanotechnology. Technologies for detecting ZIKV and DENV can be broadly divided into electrochemical, electrical, and optical. In this review, advances in DNA-nanotechnology-based ZIKV and DENV detection biosensors are discussed.
Keywords: DENV; DNA nanotechnology; ZIKV; electrical biosensor; electrochemical biosensor; optical biosensor.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Molecular and serological techniques to detect co-circulation of DENV, ZIKV and CHIKV in suspected dengue-like syndrome patients.J Clin Virol. 2016 Sep;82:108-111. doi: 10.1016/j.jcv.2016.07.017. Epub 2016 Jul 25. J Clin Virol. 2016. PMID: 27479173
-
One-step pentaplex real-time polymerase chain reaction assay for detection of zika, dengue, chikungunya, West nile viruses and a human housekeeping gene.J Clin Virol. 2019 Nov;120:44-50. doi: 10.1016/j.jcv.2019.08.011. Epub 2019 Sep 16. J Clin Virol. 2019. PMID: 31557664 Free PMC article.
-
Validation of serological and molecular methods for diagnosis of zika virus infections.J Virol Methods. 2019 Jan;263:68-74. doi: 10.1016/j.jviromet.2018.10.011. Epub 2018 Oct 18. J Virol Methods. 2019. PMID: 30342068
-
Modulation of Dengue/Zika Virus Pathogenicity by Antibody-Dependent Enhancement and Strategies to Protect Against Enhancement in Zika Virus Infection.Front Immunol. 2018 Apr 23;9:597. doi: 10.3389/fimmu.2018.00597. eCollection 2018. Front Immunol. 2018. PMID: 29740424 Free PMC article. Review.
-
Immune Response to Dengue and Zika.Annu Rev Immunol. 2018 Apr 26;36:279-308. doi: 10.1146/annurev-immunol-042617-053142. Epub 2018 Jan 18. Annu Rev Immunol. 2018. PMID: 29345964 Free PMC article. Review.
Cited by
-
Current research status of tumor cell biomarker detection.Microsyst Nanoeng. 2023 Oct 5;9:123. doi: 10.1038/s41378-023-00581-5. eCollection 2023. Microsyst Nanoeng. 2023. PMID: 37811123 Free PMC article. Review.
-
Portable Point-of-Care Diagnosis Platforms and Emerging Predictive Biomarkers for Rapid Detection of Severe Dengue Viral Infection.ACS Sens. 2025 May 23;10(5):3302-3316. doi: 10.1021/acssensors.5c00263. Epub 2025 Mar 31. ACS Sens. 2025. PMID: 40165016 Free PMC article. Review.
-
Advancing Topoisomerase Research Using DNA Nanotechnology.Small Methods. 2025 Jun;9(6):e2401113. doi: 10.1002/smtd.202401113. Epub 2024 Nov 11. Small Methods. 2025. PMID: 39526512 Free PMC article. Review.
-
Biosensing Systems Based on Graphene Oxide Fluorescence Quenching Effect.Micromachines (Basel). 2023 Jul 28;14(8):1522. doi: 10.3390/mi14081522. Micromachines (Basel). 2023. PMID: 37630058 Free PMC article. Review.
-
A one-pot method for universal Dengue virus detection by combining RT-RPA amplification and CRISPR/Cas12a assay.BMC Microbiol. 2025 Mar 25;25(1):163. doi: 10.1186/s12866-025-03882-z. BMC Microbiol. 2025. PMID: 40128655 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials

