Correlation between the Biodistribution of Bovine Leukemia Virus in the Organs and the Proviral Load in the Peripheral Blood during Early Stages of Experimentally Infected Cattle
- PMID: 36678478
- PMCID: PMC9867250
- DOI: 10.3390/pathogens12010130
Correlation between the Biodistribution of Bovine Leukemia Virus in the Organs and the Proviral Load in the Peripheral Blood during Early Stages of Experimentally Infected Cattle
Abstract
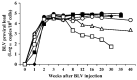
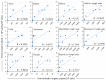
Bovine leukemia virus (BLV) is the etiological agent of enzootic bovine leukosis. However, the propagation and distribution of BLV after primary infection still need to be fully elucidated. Here, we experimentally infected seven cattle with BLV and analyzed the BLV proviral load (PVL) in the blood and various organs. BLV was first detected in the blood of the cattle after one week, and the blood PVL increased for three weeks after infection. The PVL was maintained at a high level in five cattle, while it decreased to a low or medium level in two cattle. BLV was distributed in various organs, such as the heart, lung, liver, kidney, abomasum, and thymus, and, notably, in the spleen and lymph nodes. In cattle with a high blood PVL, BLV was detected in organs other than the spleen and lymph nodes, whereas in those with a low blood PVL, BLV was only detected in the spleen and lymph nodes. The amount of BLV in the organs was comparable to that in the blood. Our findings point to the possibility of estimating the distribution of BLV provirus in organs, lymph nodes, and body fluids by measuring the blood PVL, as it was positively correlated with the biodistribution of BLV provirus in the body of BLV infection during early stages.
Keywords: BLV-CoCoMo-qPCR-2; biodistribution; bovine leukemia virus; experimentally infected cattle; organ; peripheral blood; proviral load.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

Similar articles
-
Estimation of bovine leukemia virus (BLV) proviral load harbored by lymphocyte subpopulations in BLV-infected cattle at the subclinical stage of enzootic bovine leucosis using BLV-CoCoMo-qPCR.BMC Vet Res. 2013 May 4;9:95. doi: 10.1186/1746-6148-9-95. BMC Vet Res. 2013. PMID: 23641811 Free PMC article.
-
Expression-based analysis of genes related to single nucleotide polymorphism hits associated with bovine leukemia virus proviral load in Argentinean dairy cattle.J Dairy Sci. 2021 Feb;104(2):1993-2007. doi: 10.3168/jds.2020-18924. Epub 2020 Nov 25. J Dairy Sci. 2021. PMID: 33246606
-
Risk factors associated with increased bovine leukemia virus proviral load in infected cattle in Japan from 2012 to 2014.Virus Res. 2015 Dec 2;210:283-90. doi: 10.1016/j.virusres.2015.08.020. Epub 2015 Aug 29. Virus Res. 2015. PMID: 26321160
-
Invited review: Bovine leukemia virus-Transmission, control, and eradication.J Dairy Sci. 2021 Jun;104(6):6358-6375. doi: 10.3168/jds.2020-18925. Epub 2021 Mar 23. J Dairy Sci. 2021. PMID: 33741150 Review.
-
Detrimental effect of bovine leukemia virus (BLV) on the immunological state of cattle.Vet Immunol Immunopathol. 1996 Nov;54(1-4):293-302. doi: 10.1016/s0165-2427(96)05706-6. Vet Immunol Immunopathol. 1996. PMID: 8988875 Review.
Cited by
-
BoLA-DRB3 Polymorphism Associated with Bovine Leukemia Virus Infection and Proviral Load in Holstein Cattle in Egypt.Pathogens. 2023 Dec 14;12(12):1451. doi: 10.3390/pathogens12121451. Pathogens. 2023. PMID: 38133334 Free PMC article.
-
Development of Dry and Liquid Duplex Reagent Mix-Based Polymerase Chain Reaction Assays as Novel Tools for the Rapid and Easy Quantification of Bovine Leukemia Virus (BLV) Proviral Loads.Viruses. 2024 Jun 25;16(7):1016. doi: 10.3390/v16071016. Viruses. 2024. PMID: 39066179 Free PMC article.
References
-
- Kettmann R., Deschamps J., Cleuter Y., Couez D., Burny A., Marbaix G. Leukemogenesis by bovine leukemia virus: Proviral DNA integration and lack of RNA expression of viral long terminal repeat and 3′ proximate cellular sequences. Proc. Natl. Acad. Sci. USA. 1982;79:2465–2469. doi: 10.1073/pnas.79.8.2465. - DOI - PMC - PubMed
-
- Gillet N., Florins A., Boxus M., Burteau C., Nigro A., Vandermeers F., Balon H., Bouzar A.B., Defoiche J., Burny A., et al. Mechanisms of leukemogenesis induced by bovine leukemia virus: Prospects for novel anti-retroviral therapies in human. Retrovirology. 2007;4:18. doi: 10.1186/1742-4690-4-18. - DOI - PMC - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources

