Comparing the Variants of Iron Oxide Nanoparticle-Mediated Delivery of miRNA34a for Efficiency in Silencing of PD-L1 Genes in Cancer Cells
- PMID: 36678844
- PMCID: PMC9865708
- DOI: 10.3390/pharmaceutics15010215
Comparing the Variants of Iron Oxide Nanoparticle-Mediated Delivery of miRNA34a for Efficiency in Silencing of PD-L1 Genes in Cancer Cells
Abstract
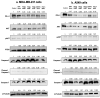
The blocking of programmed death-ligand 1 (PD-L1) in tumor cells represents a powerful strategy in cancer immunotherapy. Using viral vectors to deliver the cargo for inactivating the PD-L1 gene could be associated with host cell genotoxicity and concomitant immune attack. To develop an alternative safe gene delivery method, we designed a unique combination for miRNA34a delivery using a transgene carrier in the form of iron oxide magnetic nanoparticles (IONPs) via magnetofection to downregulate PD-L1 expression in cancer cells. We synthesized IONPs of multiple shapes (IONRs (iron oxide nanorods), IONSs (iron oxide nanospheres), and ITOHs (iron oxide truncated octahedrons)), surface-functionalized with polyethyleneimine (PEI) using the ligand exchange method, as gene delivery systems. Under the guidance of an external magnetic field, PEI@IONPs loaded with plasmid DNA (DNA/PEI@IONPs) encoding GFP showed high transfection efficiency at different weight ratios and time points in A549 and MDA-MB-231 cells. Additionally, the DNA/PEI@IONPs with miRNA34a inserts under a static magnetic field resulted in significant knockdown of the PD-L1 gene, as demonstrated via immunoblotting of the PD-L1 protein. Among the three shapes of IONPs, IONRs showed the highest PD-L1 knockdown efficiency. The genetic expression of miRNA34a was also studied using qPCR and it showed high expression of miRNA in cells treated with PEI@IONRs. Flow cytometry and a live/dead assay confirmed apoptosis after transfection with miRNA34a. To conclude, in this paper, a promising transgene carrier with low cost, negligible cytotoxicity, and high transfection efficiency has been successfully established for miRNA gene delivery in the context of cancer immunotherapy.
Keywords: PD-L1 gene; immunotherapy; iron oxide nanoparticles; miRNA34a; transfection efficiency.
Conflict of interest statement
The authors declare no conflict of interest.
Figures






References
-
- Maggiora G., Gokhale V. Non-specificity of drug-target interactions–consequences for drug discovery; Proceedings of the Frontiers in Molecular Design and Chemical Information Science-Herman Skolnik Award Symposium 2015; Boston, MA, USA. 5 October 2016; pp. 91–142.
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

