Mutational analysis of microsatellite-stable gastrointestinal cancer with high tumour mutational burden: a retrospective cohort study
- PMID: 36681091
- PMCID: PMC10599647
- DOI: 10.1016/S1470-2045(22)00783-5
Mutational analysis of microsatellite-stable gastrointestinal cancer with high tumour mutational burden: a retrospective cohort study
Abstract
Background: Genomic signatures contributing to high tumour mutational burden (TMB-H) independent from mismatch-repair deficiency (dMMR) or microsatellite instability-high (MSI-H) status are not well studied. We aimed to characterise molecular features of microsatellite stable (MSS) TMB-H gastrointestinal tumours.
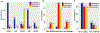
Methods: Molecular alterations of 48 606 gastrointestinal tumours from Caris Life Sciences (CARIS) identified with next-generation sequencing were compared among MSS-TMB-H, dMMR/MSI-H, and MSS-TMB-low (L) tumours, using χ2 or Fisher's exact tests. Antitumour immune response within the tumour environment was predicted by analysing the infiltration of immune cells and immune signatures using The Cancer Genome Atlas database. The Kaplan-Meier method and the log-rank test were used to evaluate the impact of gene alterations on the efficacy of immune checkpoint inhibitors in MSS gastrointestinal cancers from the CARIS database, a Memorial Sloan Kettering Cancer Center cohort, and a Peking University Cancer Hospital cohort.
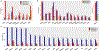
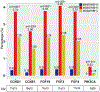
Findings: MSS-TMB-H was observed in 1600 (3·29%) of 48 606 tumours, dMMR/MSI-H in 2272 (4·67%), and MSS-TMB-L in 44 734 (92·03%). Gene mutations in SMAD2, MTOR, NFE2L2, RB1, KEAP1, TERT, and RASA1 might impair antitumour immune response despite TMB-H, while mutations in 16 other genes (CDC73, CTNNA1, ERBB4, EZH2, JAK2, MAP2K1, MAP2K4, PIK3R1, POLE, PPP2R1A, PPP2R2A, PTPN11, RAF1, RUNX1, STAG2, and XPO1) were related to TMB-H with enhanced antitumour immune response independent of dMMR/MSI-H, constructing a predictive model (modified TMB [mTMB]) for immune checkpoint inhibitor efficacy. Patients with any mutation in the mTMB gene signature, in comparison with patients with mTMB wildtype tumours, showed a superior survival benefit from immune checkpoint inhibitors in MSS gastrointestinal cancers in the CARIS cohort (n=95, median overall survival 18·77 months [95% CI 17·30-20·23] vs 7·03 months [5·73-8·34]; hazard ratio 0·55 [95% CI 0·31-0·99], p=0·044). In addition, copy number amplification in chromosome 11q13 (eg, CCND1, FGF genes) was more prevalent in MSS-TMB-H tumours than in the dMMR/MSI-H or MSS-TMB-L subgroups.
Interpretation: Not all mutations related to TMB-H can enhance antitumour immune response. More composite biomarkers should be investigated (eg, mTMB signature) to tailor treatment with immune checkpoint inhibitors. Our data also provide novel insights for the combination of immune checkpoint inhibitors and drugs targeting cyclin D1 or FGFs.
Funding: US National Cancer Institute, Gloria Borges WunderGlo Foundation, Dhont Family Foundation, Gene Gregg Pancreas Research Fund, San Pedro Peninsula Cancer Guild, Daniel Butler Research Fund, Victoria and Philip Wilson Research Fund, Fong Research Project, Ming Hsieh Research Fund, Shanghai Sailing Program, China National Postdoctoral Program for Innovative Talents, China Postdoctoral Science Foundation, National Natural Science Foundation of China.
Copyright © 2023 Elsevier Ltd. All rights reserved.
Conflict of interest statement
Declaration of interests H-JL reports receiving honoraria from serving as a consultant or from advisory board membership for Merck Serono, Bayer, and Genentech. JX, AF, YB, MO, DS, and WMK are employees of Caris Life Sciences. AFS reports funding for research, travel, and speakers bureau participation from Caris Life Sciences. BAW reports receiving honoraria from Bayer, Sirtex, Lilly, Taiho, and HalioDx. All other authors declare no competing interests.
Figures
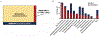

References
-
- Marabelle A, Fakih M, Lopez J, et al. Association of tumour mutational burden with outcomes in patients with advanced solid tumours treated with pembrolizumab: prospective biomarker analysis of the multicohort, open-label, phase 2 KEYNOTE-158 study. The Lancet Oncology 2020; 21(10): 1353–65. - PubMed
-
- Wang F, Wei XL, Wang FH, et al. Safety, efficacy and tumor mutational burden as a biomarker of overall survival benefit in chemo-refractory gastric cancer treated with toripalimab, a PD-1 antibody in phase Ib/II clinical trial NCT02915432. Ann Oncol 2019; 30(9): 1479–86. - PMC - PubMed
-
- Kim ST, Cristescu R, Bass AJ, et al. Comprehensive molecular characterization of clinical responses to PD-1 inhibition in metastatic gastric cancer. Nat Med 2018; 24(9): 1449–58. - PubMed
-
- Fukuoka S, Hara H, Takahashi N, et al. Regorafenib Plus Nivolumab in Patients With Advanced Gastric or Colorectal Cancer: An Open-Label, Dose-Escalation, and Dose-Expansion Phase Ib Trial (REGONIVO, EPOC1603). J Clin Oncol 2020; 38(18): 2053–61. - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

