Safety, Tolerability and Pharmacokinetics of Half-Life Extended Severe Acute Respiratory Syndrome Coronavirus 2 Neutralizing Monoclonal Antibodies AZD7442 (Tixagevimab-Cilgavimab) in Healthy Adults
- PMID: 36683419
- PMCID: PMC10175065
- DOI: 10.1093/infdis/jiad014
Safety, Tolerability and Pharmacokinetics of Half-Life Extended Severe Acute Respiratory Syndrome Coronavirus 2 Neutralizing Monoclonal Antibodies AZD7442 (Tixagevimab-Cilgavimab) in Healthy Adults
Abstract
Background: AZD7442 is a combination of extended half-life, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2)-specific neutralizing monoclonal antibodies (tixagevimab and cilgavimab).
Methods: This phase 1, first-in-human, randomized, double-blind, placebo-controlled, dose-escalation study evaluated AZD7442 administered intramuscularly (300 mg) or intravenously (300, 1000, or 3000 mg) in healthy adults (aged 18-55 years). The primary end point was safety and tolerability. Secondary end points included pharmacokinetics and antidrug antibodies.
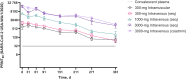
Results: Between 18 August and 16 October 2020, a total of 60 participants were enrolled; 50 received AZD7442, and 10 received placebo. Adverse events (all of mild or moderate intensity) occurred in 26 participants (52.0%) in the AZD7442 groups and 8 (80.0%) in the placebo group. No infusion or injection site or hypersensitivity reactions occurred. Tixagevimab and cilgavimab had mean half-lives of approximately 90 days (range, 87.0-95.3 days for tixagevimab and 79.8--91.1 days for cilgavimab) and similar pharmacokinetic profiles over the 361-day study period. SARS-CoV-2-specific neutralizing antibody titers provided by AZD7442 were maintained above those in plasma from convalescent patients with coronavirus disease 2019 (COVID-19).
Conclusions: AZD7442 was well tolerated in healthy adults, showing a favorable safety profile across all doses. Depending on the SARS-CoV-2 variant, pharmacokinetic analyses suggest the AZD7442 could offer protection for ≥6 months against symptomatic COVID-19 after a single 300-mg intramuscular administration.
Clinical trials registration: NCT04507256.
Keywords: COVID-19; SARS-CoV-2; monoclonal antibody; pharmacokinetics; phase 1; safety; tolerability.
Plain language summary
Antibodies are proteins produced by the body in response to infections caused by microbes, including viruses. AZD7442 is a combination of 2 human antibodies, with an extended duration of effect, sourced from people who had recovered from coronavirus disease 2019 (COVID-19). These antibodies recognize a specific part (spike protein) of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the virus that causes COVID-19, and prevent the virus from infecting cells in the body. The current study evaluated the safety of AZD7442 in healthy volunteers. Sixty adults were given AZD7442 or placebo (salt solution) as injections into the muscle (300-mg dose) or infusions into a vein (300–3000-mg doses). The study did not find any safety issues with AZD7442, including at the highest dose. AZD7442 was measured in the blood 12 months after dosing, suggesting a long duration of protection. Following this study, AZD7442 was tested in larger clinical trials to investigate its potential in preventing and treating COVID-19. AZD7442 is currently authorized as treatment for outpatients with COVID-19 and as a preventive drug in people who may not respond well to COVID-19 vaccines and need additional protection (eg, those taking medications that dampen the immune system).
© The Author(s) 2023. Published by Oxford University Press on behalf of Infectious Diseases Society of America.
Conflict of interest statement
Potential conflicts of interest. P. F. S. is an employee of Parexel International and does not hold stock in AstraZeneca. M. A., D. B., R. H. A., J. T., A. A. A., J. B., C. C., A. G., R. J. K., V. P. R., S. S., K. S., A. T., and M. T. E. were employees of, and hold or may have held stock in, AstraZeneca at the time of study. All authors have submitted the ICMJE Form for Disclosure of Potential Conflicts of Interest. Conflicts that the editors consider relevant to the content of the manuscript have been disclosed.
Figures


Similar articles
-
Safety, Efficacy and Pharmacokinetics of AZD7442 (Tixagevimab/Cilgavimab) for Treatment of Mild-to-Moderate COVID-19: 15-Month Final Analysis of the TACKLE Trial.Infect Dis Ther. 2024 Mar;13(3):521-533. doi: 10.1007/s40121-024-00931-4. Epub 2024 Feb 25. Infect Dis Ther. 2024. PMID: 38403865 Free PMC article.
-
Safety, tolerability, and pharmacokinetics of half-life extended SARS-CoV-2-neutralizing monoclonal antibodies AZD7442 (tixagevimab/cilgavimab) in healthy Japanese adults.J Infect Chemother. 2023 Nov;29(11):1061-1067. doi: 10.1016/j.jiac.2023.07.014. Epub 2023 Jul 29. J Infect Chemother. 2023. PMID: 37524201 Clinical Trial.
-
Efficacy, Safety, and Pharmacokinetics of AZD7442 (Tixagevimab/Cilgavimab) for Prevention of Symptomatic COVID-19: 15-Month Final Analysis of the PROVENT and STORM CHASER Trials.Infect Dis Ther. 2024 Jun;13(6):1253-1268. doi: 10.1007/s40121-024-00970-x. Epub 2024 May 4. Infect Dis Ther. 2024. PMID: 38703336 Free PMC article.
-
Tixagevimab/cilgavimab for prevention and treatment of COVID-19: a review.Expert Rev Anti Infect Ther. 2022 Dec;20(12):1517-1527. doi: 10.1080/14787210.2022.2134118. Epub 2022 Oct 27. Expert Rev Anti Infect Ther. 2022. PMID: 36217836 Review.
-
A Critical Analysis of the Use of Cilgavimab plus Tixagevimab Monoclonal Antibody Cocktail (Evusheld™) for COVID-19 Prophylaxis and Treatment.Viruses. 2022 Sep 9;14(9):1999. doi: 10.3390/v14091999. Viruses. 2022. PMID: 36146805 Free PMC article. Review.
Cited by
-
Antiviral effect of Evusheld in COVID-19 hospitalized patients infected with pre-Omicron or Omicron variants: a modelling analysis of the randomized DisCoVeRy trial.J Antimicrob Chemother. 2024 Nov 4;79(11):2887-2895. doi: 10.1093/jac/dkae301. J Antimicrob Chemother. 2024. PMID: 39236218 Free PMC article. Clinical Trial.
-
Long-term outcomes of passive immunotherapy for COVID-19: a pooled analysis of a large multinational platform randomized clinical trial.Clin Microbiol Infect. 2025 Jun;31(6):1053-1060. doi: 10.1016/j.cmi.2025.02.002. Epub 2025 Feb 6. Clin Microbiol Infect. 2025. PMID: 39922466 Free PMC article. Clinical Trial.
-
Serum AZD7442 (tixagevimab-cilgavimab) concentrations and in vitroIC50 values predict SARS-CoV-2 neutralising antibody titres.Clin Transl Immunology. 2024 Jun 13;13(6):e1517. doi: 10.1002/cti2.1517. eCollection 2024. Clin Transl Immunology. 2024. PMID: 38873124 Free PMC article.
-
Safety and Efficacy of Combined Tixagevimab and Cilgavimab Administered Intramuscularly or Intravenously in Nonhospitalized Patients With COVID-19: 2 Randomized Clinical Trials.JAMA Netw Open. 2023 Apr 3;6(4):e2310039. doi: 10.1001/jamanetworkopen.2023.10039. JAMA Netw Open. 2023. PMID: 37099295 Free PMC article. Clinical Trial.
-
Safety, Efficacy and Pharmacokinetics of AZD7442 (Tixagevimab/Cilgavimab) for Treatment of Mild-to-Moderate COVID-19: 15-Month Final Analysis of the TACKLE Trial.Infect Dis Ther. 2024 Mar;13(3):521-533. doi: 10.1007/s40121-024-00931-4. Epub 2024 Feb 25. Infect Dis Ther. 2024. PMID: 38403865 Free PMC article.
References
-
- Haas EJ, Angulo FJ, McLaughlin JM, et al. . Impact and effectiveness of mRNA BNT162b2 vaccine against SARS-CoV-2 infections and COVID-19 cases, hospitalisations, and deaths following a nationwide vaccination campaign in Israel: an observational study using national surveillance data. Lancet 2021; 397:1819–29. - PMC - PubMed
Publication types
MeSH terms
Substances
Supplementary concepts
Associated data
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

