Microglia-Mediated Neurovascular Unit Dysfunction in Alzheimer's Disease
- PMID: 36683511
- PMCID: PMC10473143
- DOI: 10.3233/JAD-221064
Microglia-Mediated Neurovascular Unit Dysfunction in Alzheimer's Disease
Abstract
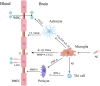
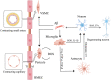
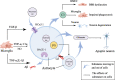
The neurovascular unit (NVU) is involved in the pathological changes in Alzheimer's disease (AD). The NVU is a structural and functional complex that maintains microenvironmental homeostasis and metabolic balance in the central nervous system. As one of the most important components of the NVU, microglia not only induce blood-brain barrier breakdown by promoting neuroinflammation, the infiltration of peripheral white blood cells and oxidative stress but also mediate neurovascular uncoupling by inducing mitochondrial dysfunction in neurons, abnormal contraction of cerebral vessels, and pericyte loss in AD. In addition, microglia-mediated dysfunction of cellular components in the NVU, such as astrocytes and pericytes, can destroy the integrity of the NVU and lead to NVU impairment. Therefore, we review the mechanisms of microglia-mediated NVU dysfunction in AD. Furthermore, existing therapeutic advancements aimed at restoring the function of microglia and the NVU in AD are discussed. Finally, we predict the role of pericytes in microglia-mediated NVU dysfunction in AD is the hotspot in the future.
Keywords: Alzheimer’s disease; blood-brain barrier; microglia; neurovascular uncoupling; neurovascular unit; pericyte.
Conflict of interest statement
The authors have no conflict of interest to report.
Figures
References
Publication types
MeSH terms
LinkOut - more resources
Full Text Sources
Medical

