Acute infection with Brachyspira hyodysenteriae affects mucin expression, glycosylation, and fecal MUC5AC
- PMID: 36683692
- PMCID: PMC9852840
- DOI: 10.3389/fcimb.2022.1042815
Acute infection with Brachyspira hyodysenteriae affects mucin expression, glycosylation, and fecal MUC5AC
Abstract
Introduction: Infection with strongly β-hemolytic strains of Brachyspira hyodysenteriae leads to swine dysentery (SD), a production-limiting disease that causes mucohemorrhagic diarrhea and typhlocolitis in pigs. This pathogen has strong chemotactic activity toward mucin, and infected pigs often have a disorganized mucus layer and marked de novo expression of MUC5AC, which is not constitutively expressed in the colon. It has been shown that fucose is chemoattractant for B. hyodysenteriae, and a highly fermentable fiber diet can mitigate and delay the onset of SD.
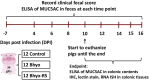
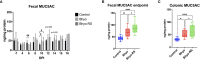
Methods: We used lectins targeting sialic acids in α-2,6 or α-2,3 linkages, N-acetylglucosamine (GlcNAc), α-linked L-fucose, and an immunohistochemical stain targeting N-glycolylneuraminic acid (NeuGc) to investigate the local expression of these mucin glycans in colonic tissues of pigs with acute SD. We used a commercial enzyme-linked immunosorbent assay (ELISA) to quantify fecal MUC5AC in infected pigs and assess its potential as a diagnostic monitoring tool and RNA in situ hybridization to detect IL-17A in the colonic mucosa.
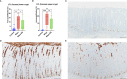
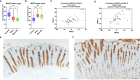
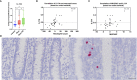
Results: Colonic mucin glycosylation during SD has an overall increase in fucose, a spatially different distribution of GlcNAc with more expression within the crypt lumens of the upper colonic mucosa, and decreased expression or a decreased trend of sialic acids in α-2,6 or α-2,3 linkages, and NeuGc compared to the controls. The degree of increased fucosylation was less in the colonic mucosa of pigs with SD and fed the highly fermentable fiber diet. There was a significant increase in MUC5AC in fecal and colonic samples of pigs with SD at the endpoint compared to the controls, but the predictive value for disease progression was limited.
Discussion: Fucosylation and the impact of dietary fiber may play important roles in the pathogenesis of SD. The lack of predictive value for fecal MUC5AC quantification by ELISA is possibly due to the presence of other non-colonic sources of MUC5AC in the feces. The moderate correlation between IL-17A, neutrophils and MUC5AC confirms its immunoregulatory and mucin stimulatory role. Our study characterizes local alteration of mucin glycosylation in the colonic mucosa of pigs with SD after B. hyodysenteriae infection and may provide insight into host-pathogen interaction.
Keywords: Brachyspira; GlcNAc; MUC2; MUC5AC; fucose; glycosylation; sialic acid; swine dysentery.
Copyright © 2023 Lin, Helm, Gabler and Burrough.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Comparison of sesion severity, distribution, and colonic mucin expression in pigs with acute swine dysentery following oral inoculation with "Brachyspira hampsonii" or Brachyspira hyodysenteriae.Vet Pathol. 2014 Nov;51(6):1096-108. doi: 10.1177/0300985813516646. Epub 2014 Feb 27. Vet Pathol. 2014. PMID: 24577722
-
Brachyspira hyodysenteriae Infection Regulates Mucin Glycosylation Synthesis Inducing an Increased Expression of Core-2 O-Glycans in Porcine Colon.J Proteome Res. 2017 Apr 7;16(4):1728-1742. doi: 10.1021/acs.jproteome.7b00002. Epub 2017 Mar 30. J Proteome Res. 2017. PMID: 28301166
-
The levels of Brachyspira hyodysenteriae binding to porcine colonic mucins differ between individuals, and binding is increased to mucins from infected pigs with de novo MUC5AC synthesis.Infect Immun. 2015 Apr;83(4):1610-9. doi: 10.1128/IAI.03073-14. Epub 2015 Feb 2. Infect Immun. 2015. PMID: 25644008 Free PMC article.
-
Serologic detection of Brachyspira (Serpulina) hyodysenteriae infections.Anim Health Res Rev. 2001 Jun;2(1):45-52. Anim Health Res Rev. 2001. PMID: 11708746 Review.
-
Swine dysentery: aetiology, pathogenicity, determinants of transmission and the fight against the disease.Int J Environ Res Public Health. 2013 May 10;10(5):1927-47. doi: 10.3390/ijerph10051927. Int J Environ Res Public Health. 2013. PMID: 23665849 Free PMC article. Review.
Cited by
-
Deep crypt secretory cells shape region-specific mucin glycosylation patterns in the mouse colon.PLoS One. 2025 Jul 8;20(7):e0326157. doi: 10.1371/journal.pone.0326157. eCollection 2025. PLoS One. 2025. PMID: 40627604 Free PMC article.
-
The formidable guardian: Type 3 immunity in the intestine of pigs.Virulence. 2024 Dec;15(1):2424325. doi: 10.1080/21505594.2024.2424325. Epub 2024 Nov 8. Virulence. 2024. PMID: 39497434 Free PMC article. Review.
-
Mucin 5AC as a Biomarker for Sessile Serrated Lesions: Results From a Systematic Review and Meta-Analysis.Clin Transl Gastroenterol. 2025 May 1;16(5):e00831. doi: 10.14309/ctg.0000000000000831. Clin Transl Gastroenterol. 2025. PMID: 40110854 Free PMC article.
-
Sialic Acid Receptor Specificity in Mammary Gland of Dairy Cattle Infected with Highly Pathogenic Avian Influenza A(H5N1) Virus.Emerg Infect Dis. 2024 Jul;30(7):1361-1373. doi: 10.3201/eid3007.240689. Epub 2024 Jun 11. Emerg Infect Dis. 2024. PMID: 38861554 Free PMC article.
-
Effects of fermented wheat bran on growth performance, nutrient digestibility and intestinal microbiota of weaned piglets.Front Vet Sci. 2025 Apr 16;12:1561196. doi: 10.3389/fvets.2025.1561196. eCollection 2025. Front Vet Sci. 2025. PMID: 40308694 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

