Inhalable microparticles as drug delivery systems to the lungs in a dry powder formulations
- PMID: 36683752
- PMCID: PMC9845529
- DOI: 10.1093/rb/rbac099
Inhalable microparticles as drug delivery systems to the lungs in a dry powder formulations
Abstract
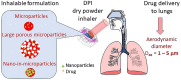
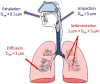
Inhalation-administrated drugs remain an interesting possibility of addressing pulmonary diseases. Direct drug delivery to the lungs allows one to obtain high concentration in the site of action with limited systemic distribution, leading to a more effective therapy with reduced required doses and side effects. On the other hand, there are several difficulties in obtaining a formulation that would meet all the criteria related to physicochemical, aerodynamic and biological properties, which is the reason why only very few of the investigated systems can reach the clinical trial phase and proceed to everyday use as a result. Therefore, we focused on powders consisting of polysaccharides, lipids, proteins or natural and synthetic polymers in the form of microparticles that are delivered by inhalation to the lungs as drug carriers. We summarized the most common trends in research today to provide the best dry powders in the right fraction for inhalation that would be able to release the drug before being removed by natural mechanisms. This review article addresses the most common manufacturing methods with novel modifications, pros and cons of different materials, drug loading capacities with release profiles, and biological properties such as cytocompatibility, bactericidal or anticancer properties.
Keywords: drug delivery systems to lungs; dry powder inhalers; inhalers; microparticles; pulmonary therapies.
© The Author(s) 2022. Published by Oxford University Press.
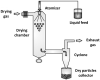
Figures






Similar articles
-
Design and evaluation of novel inhalable sildenafil citrate spray-dried microparticles for pulmonary arterial hypertension.J Control Release. 2019 May 28;302:126-139. doi: 10.1016/j.jconrel.2019.03.029. Epub 2019 Mar 30. J Control Release. 2019. PMID: 30940497
-
Preparation and evaluation of poly(lactic-co-glycolic acid) microparticles as a carrier for pulmonary delivery of recombinant human interleukin-2: II. In vitro studies on aerodynamic properties of dry powder inhaler formulations.Drug Dev Ind Pharm. 2011 Nov;37(11):1376-86. doi: 10.3109/03639045.2011.576680. Epub 2011 May 6. Drug Dev Ind Pharm. 2011. PMID: 21548727
-
Development and Evaluation of Chitosan Microparticles Based Dry Powder Inhalation Formulations of Rifampicin and Rifabutin.J Aerosol Med Pulm Drug Deliv. 2016 Apr;29(2):179-95. doi: 10.1089/jamp.2014.1187. Epub 2015 Sep 25. J Aerosol Med Pulm Drug Deliv. 2016. PMID: 26406162
-
Exploring inhalable polymeric dry powders for anti-tuberculosis drug delivery.Mater Sci Eng C Mater Biol Appl. 2018 Dec 1;93:1090-1103. doi: 10.1016/j.msec.2018.09.004. Epub 2018 Sep 11. Mater Sci Eng C Mater Biol Appl. 2018. PMID: 30274040 Review.
-
Dry powder inhalers of antitubercular drugs.Tuberculosis (Edinb). 2022 Jul;135:102228. doi: 10.1016/j.tube.2022.102228. Epub 2022 Jun 24. Tuberculosis (Edinb). 2022. PMID: 35779497 Review.
Cited by
-
Cancer cell response to extrinsic and intrinsic mechanical cue: opportunities for tumor apoptosis strategies.Regen Biomater. 2024 Feb 20;11:rbae016. doi: 10.1093/rb/rbae016. eCollection 2024. Regen Biomater. 2024. PMID: 38476678 Free PMC article. Review.
-
Microscale Delivery Systems for Hydrophilic Active Ingredients in Functional Consumer Goods.Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2025 Mar-Apr;17(2):e70009. doi: 10.1002/wnan.70009. Wiley Interdiscip Rev Nanomed Nanobiotechnol. 2025. PMID: 40223375 Free PMC article. Review.
-
Towards More Precise Targeting of Inhaled Aerosols to Different Areas of the Respiratory System.Pharmaceutics. 2024 Jan 10;16(1):97. doi: 10.3390/pharmaceutics16010097. Pharmaceutics. 2024. PMID: 38258107 Free PMC article. Review.
-
Enhancing tumor photodynamic synergistic therapy efficacy through generation of carbon radicals by Prussian blue nanomedicine.Regen Biomater. 2024 Aug 24;11:rbae103. doi: 10.1093/rb/rbae103. eCollection 2024. Regen Biomater. 2024. PMID: 39346686 Free PMC article.
-
Role of shock waves in materials processing: Fundamentals and Applications.Ultrason Sonochem. 2025 Jul 19;120:107473. doi: 10.1016/j.ultsonch.2025.107473. Online ahead of print. Ultrason Sonochem. 2025. PMID: 40706420 Free PMC article.
References
-
- ‘Drug Delivery Systems’. https://www.nibib.nih.gov/science-education/science-topics/drug-delivery... (24 March 2022, date last accessed).
-
- Pandey A. An overview on advances in the nanocarriers drug delivery systems. In: Shukla A (eds). EMR/ESR/EPR Spectroscopy Characterization of Nanomaterials, New Delhi: Springer, 2017;62:65–76. doi: 10.1007/978-81-322-3655-9_3. - DOI
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

