Pre-existing Toxoplasma gondii infection increases susceptibility to pentylenetetrazol-induced seizures independent of traumatic brain injury in mice
- PMID: 36683847
- PMCID: PMC9849700
- DOI: 10.3389/fnmol.2022.1079097
Pre-existing Toxoplasma gondii infection increases susceptibility to pentylenetetrazol-induced seizures independent of traumatic brain injury in mice
Abstract
Introduction: Post-traumatic epilepsy (PTE) is a debilitating chronic outcome of traumatic brain injury (TBI), and neuroinflammation is implicated in increased seizure susceptibility and epileptogenesis. However, how common clinical factors, such as infection, may modify neuroinflammation and PTE development has been understudied. The neurotropic parasite, Toxoplasma gondii (T. gondii) incurably infects one-third of the world's population. Thus, many TBI patients have a pre-existing T. gondii infection at the time of injury. T. gondii infection results in chronic low-grade inflammation and altered signaling pathways within the brain, and preliminary clinical evidence suggest that it may be a risk factor for epilepsy. Despite this, no studies have considered how a pre-existing T. gondii infection may alter the development of PTE.
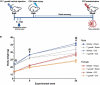
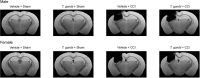
Methods: This study aimed to provide insight into this knowledge gap by assessing how a pre-existing T. gondii infection alters susceptibility to, and severity of, pentylenetetrazol (PTZ)-induced seizures (i.e., a surrogate marker of epileptogenesis/PTE) at a chronic stage of TBI recovery. We hypothesized that T. gondii will increase the likelihood and severity of seizures following PTZ administration, and that this would occur in the presence of intensified neuroinflammation. To test this, 6-week old male and female C57BL/6 Jax mice were intraperitoneally injected with 50,000 T. gondii tachyzoites or with the PBS vehicle only. At 12-weeks old, mice either received a severe TBI via controlled cortical impact or sham injury. At 18-weeks post-injury, mice were administered 40 mg/kg PTZ and video-recorded for evaluation of seizure susceptibility. Fresh cortical tissue was then collected for gene expression analyses.
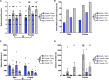
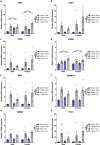
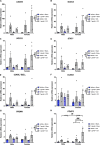
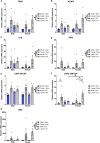
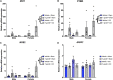
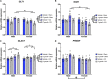
Results: Although no synergistic effects were evident between infection and TBI, chronic T. gondii infection alone had robust effects on the PTZ-seizure response and gene expression of markers related to inflammatory, oxidative stress, and glutamatergic pathways. In addition to this, females were more susceptible to PTZ-induced seizures than males. While TBI did not impact PTZ responses, injury effects were evident at the molecular level.
Discussion: Our data suggests that a pre-existing T. gondii infection is an important modifier of seizure susceptibility independent of brain injury, and considerable attention should be directed toward delineating the mechanisms underlying this pro-epileptogenic factor.
Keywords: epileptogenesis; immune response; neuroinflammation; oxidative stress; post-traumatic epilepsy (PTE).
Copyright © 2023 Baker, Uboldi, Tonkin, Wright, Vo, Wilson, Mychasiuk, McDonald, Semple, Sun and Shultz.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
References
-
- Bergersen K. V., Barnes A., Worth D., David C., Wilson E. H. (2021). Targeted transcriptomic analysis of C57BL/6 And BALB/c mice during progressive chronic Toxoplasma gondii infection reveals changes in host and parasite gene expression relating to neuropathology and resolution. Front. Cell. Infect. Microbiol. 11:645778. 10.3389/fcimb.2021.645778 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

