Guanylyl cyclase/natriuretic peptide receptor-A: Identification, molecular characterization, and physiological genomics
- PMID: 36683859
- PMCID: PMC9846370
- DOI: 10.3389/fnmol.2022.1076799
Guanylyl cyclase/natriuretic peptide receptor-A: Identification, molecular characterization, and physiological genomics
Abstract
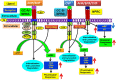
The natriuretic peptides (NPs) hormone family, which consists mainly of atrial, brain, and C-type NPs (ANP, BNP, and CNP), play diverse roles in mammalian species, ranging from renal, cardiac, endocrine, neural, and vascular hemodynamics to metabolic regulations, immune responsiveness, and energy distributions. Over the last four decades, new data has transpired regarding the biochemical and molecular compositions, signaling mechanisms, and physiological and pathophysiological functions of NPs and their receptors. NPs are incremented mainly in eliciting natriuretic, diuretic, endocrine, vasodilatory, and neurological activities, along with antiproliferative, antimitogenic, antiinflammatory, and antifibrotic responses. The main locus responsible in the biological and physiological regulatory actions of NPs (ANP and BNP) is the plasma membrane guanylyl cyclase/natriuretic peptide receptor-A (GC-A/NPRA), a member of the growing multi-limbed GC family of receptors. Advances in this field have provided tremendous insights into the critical role of Npr1 (encoding GC-A/NPRA) in the reduction of fluid volume and blood pressure homeostasis, protection against renal and cardiac remodeling, and moderation and mediation of neurological disorders. The generation and use of genetically engineered animals, including gene-targeted (gene-knockout and gene-duplication) and transgenic mutant mouse models has revealed and clarified the varied roles and pleiotropic functions of GC-A/NPRA in vivo in intact animals. This review provides a chronological development of the biochemical, molecular, physiological, and pathophysiological functions of GC-A/NPRA, including signaling pathways, genomics, and gene regulation in both normal and disease states.
Keywords: cyclic-GMP signaling; gene-targeting; genomics; natriuretic peptides; particulate guanylyl cyclase receptor-A; structure–function relationship.
Copyright © 2023 Pandey.
Conflict of interest statement
The author declares that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Molecular and genetic aspects of guanylyl cyclase natriuretic peptide receptor-A in regulation of blood pressure and renal function.Physiol Genomics. 2018 Nov 1;50(11):913-928. doi: 10.1152/physiolgenomics.00083.2018. Epub 2018 Aug 31. Physiol Genomics. 2018. PMID: 30169131 Free PMC article. Review.
-
Molecular Signaling Mechanisms and Function of Natriuretic Peptide Receptor-A in the Pathophysiology of Cardiovascular Homeostasis.Front Physiol. 2021 Aug 19;12:693099. doi: 10.3389/fphys.2021.693099. eCollection 2021. Front Physiol. 2021. PMID: 34489721 Free PMC article. Review.
-
The functional genomics of guanylyl cyclase/natriuretic peptide receptor-A: perspectives and paradigms.FEBS J. 2011 Jun;278(11):1792-807. doi: 10.1111/j.1742-4658.2011.08081.x. Epub 2011 Apr 7. FEBS J. 2011. PMID: 21375691 Free PMC article. Review.
-
Intracellular trafficking and metabolic turnover of ligand-bound guanylyl cyclase/atrial natriuretic peptide receptor-A into subcellular compartments.Mol Cell Biochem. 2002 Jan;230(1-2):61-72. Mol Cell Biochem. 2002. PMID: 11952097 Review.
-
Guanylyl cyclase/natriuretic peptide receptor-A signaling antagonizes phosphoinositide hydrolysis, Ca(2+) release, and activation of protein kinase C.Front Mol Neurosci. 2014 Aug 22;7:75. doi: 10.3389/fnmol.2014.00075. eCollection 2014. Front Mol Neurosci. 2014. PMID: 25202235 Free PMC article. Review.
References
-
- Arise K. K., Kumar P., Garg R., Samivel R., Zhao H., Pandya K., et al. . (2020). Angiotensin II represses Npr1 expression and receptor function by recruitment of transcription factors CREB and HSF-4a and activation of HDACs. Sci. Rep. 10:4337. doi: 10.1038/s41598-020-61041-y, PMID: - DOI - PMC - PubMed
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

