Identification and functional studies of microbial volatile organic compounds produced by Arctic flower yeasts
- PMID: 36684747
- PMCID: PMC9850290
- DOI: 10.3389/fpls.2022.941929
Identification and functional studies of microbial volatile organic compounds produced by Arctic flower yeasts
Abstract
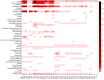
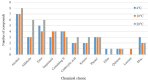
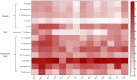
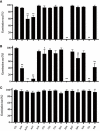
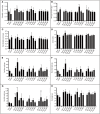
Microbial volatile organic compounds (mVOCs) can serve as a communication channel among microorganisms, insects and plants, making them important in ecosystem. In order to understand the possible role of mVOCs in Arctic ecology, the microbes in Arctic flowers and their mVOCs and effects on plants were investigated. This study aims to isolate different yeast species from the flowers of five Arctic plant species and further to explore the function of mVOCs emitted by these microbes to plant. It was found that the composition and amount of mVOCs produced by the isolated yeasts were considerably affected by changes in incubation temperature. When the incubation temperature rose, the species of alcohols, aldehydes, esters, organic acids, and ketones increased, but substances specific to low temperature decreased or disappeared. When yeasts were co-cultured with Arabidopsis thaliana without any direct contact, mVOCs produced by the isolated yeasts inhibited the seed germination of A. thaliana at low temperatures; however, the mVOCs promoted the chlorophyll content, fresh weight, root weight and flowering rate of Arabidopsis plants. Although the overall growth-promoting effect of yeast mVOCs was higher at 20°C than at 10°C, the growth-promoting effect on roots, flowers and chlorophyll was highest at 10°C. When cultured at 10°C, the mVOCs produced by Cystofilobasidium capitatum A37, Cryptococcus sp. D41, and Sporidiobolus salmonicolor D27 had the highest growth-promoting effects on the root, flowering rate and chlorophyll content of Arabidopsis, respectively. In the co-culture system, some new mVOCs were detected, such as hendecane, tetradecane, and 1-hexanol that have been proven to promote plant growth. In addition, mVOCs of the isolated Arctic yeasts could inhibit the growth of several microorganisms, especially filamentous fungi. It was the first time to prove that mVOCs produced by the isolated yeasts had varying effects on plant growth at different incubating temperatures, providing a reference for the interactions between microorganisms and plants and their possible responses to climate change in the Arctic area. Moreover, the characteristics of promoting plant growth and inhibiting microbial growth by mVOCs of Arctic yeasts would lay a foundation for potential applications in the future.
Keywords: Arctic; MVOC; flower; nectar; yeasts.
Copyright © 2023 Niu, Li, Zhang, Yao, Zhang, Liu, Peng, Huang and Peng.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


References
-
- Becher P. G., Flick G., Rozpędowska E., Schmidt A., Hagman A., Lebreton S., et al. (2012). Yeast, not fruit volatiles mediate drosophila melanogaster attraction, oviposition and development. Funct. Ecol. 26 (4), 822–828. doi: 10.1111/j.1365-2435.2012.02006.x - DOI
-
- Blom D., Fabbri C., Connor E. C., Schiestl F. P., Klauser D. R., Boller T., et al. (2011). Production of plant growth modulating volatiles is widespread among rhizosphere bacteria and strongly depends on culture conditions. Environ. Microbiol. 13 (11), 3047–3058. doi: 10.1111/j.1462-2920.2011.02582.x - DOI - PubMed
LinkOut - more resources
Full Text Sources

