Holothuria scabra extracts confer neuroprotective effect in C. elegans model of Alzheimer's disease by attenuating amyloid-β aggregation and toxicity
- PMID: 36685078
- PMCID: PMC9845652
- DOI: 10.1016/j.jtcme.2022.10.006
Holothuria scabra extracts confer neuroprotective effect in C. elegans model of Alzheimer's disease by attenuating amyloid-β aggregation and toxicity
Abstract
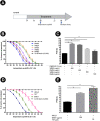
Background and aim: Alzheimer's disease (AD) is the most common aged-related neurodegenerative disorder that is associated with the toxic amyloid-β (Aβ) aggregation in the brain. While the efficacies of available drugs against AD are still limited, natural products have been shown to possess neuroprotective potential for prevention and therapy of AD. This study aimed to investigate the neuroprotective effects of H. scabra extracts against Aβ aggregation and proteotoxicity in C. elegans model of Alzheimer's diseases.
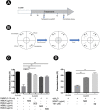
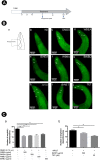
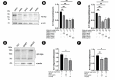
Experimental procedure: Whole bodies (WB) and body wall (BW) of H. scabra were extracted and fractionated into ethyl acetate (WBEA, BWEA), butanol (WBBU, BWBU), and ethanol (BWET). Then C. elegans AD models were treated with these fractions and investigated for Aβ aggregation and polymerization, biochemical and behavioral changes, and level of oxidative stress, as well as lifespan extension.
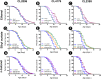
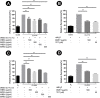
Results and conclusion: C. elegans AD model treated with H. scabra extracts, especially triterpene glycoside-rich ethyl acetate and butanol fractions, exhibited significant reduction of Aβ deposition. These H. scabra extracts also attenuated the paralysis behavior and improved the neurological defects in chemotaxis caused by Aβ aggregation. Immunoblot analysis revealed decreased level of Aβ oligomeric forms and the increased level of Aβ monomers after treatments with H. scabra extracts. In addition, H. scabra extracts reduced reactive oxygen species and increased the mean lifespan of the treated AD worms. In conclusion, this study demonstrated strong evidence of anti-Alzheimer effects by H. scabra extracts, implying that these extracts can potentially be applied as natural preventive and therapeutic agents for AD.
Taxonomy classification by evise: Alzheimer's disease, Neurodegenerative disorder, Traditional medicine, Experimental model systems, Molecular biology.
Keywords: Alzheimer's disease; Amyloid-β; Caenorhabditis elegans; Neuroprotection; Sea cucumber.
© 2022 Center for Food and Biomolecules, National Taiwan University. Production and hosting by Elsevier Taiwan LLC.
Conflict of interest statement
The authors report no conflict of interest in this study.
Figures


Similar articles
-
Holothuria scabra extracts possess anti-oxidant activity and promote stress resistance and lifespan extension in Caenorhabditis elegans.Exp Gerontol. 2018 Sep;110:158-171. doi: 10.1016/j.exger.2018.06.006. Epub 2018 Jun 11. Exp Gerontol. 2018. PMID: 29902502
-
Holothuria scabra extracts exhibit anti-Parkinson potential in C. elegans: A model for anti-Parkinson testing.Nutr Neurosci. 2018 Jul;21(6):427-438. doi: 10.1080/1028415X.2017.1299437. Epub 2017 Mar 9. Nutr Neurosci. 2018. PMID: 28276260
-
Beneficial Effects of Cyclic Ether 2-Butoxytetrahydrofuran from Sea Cucumber Holothuria scabra against Aβ Aggregate Toxicity in Transgenic Caenorhabditis elegans and Potential Chemical Interaction.Molecules. 2021 Apr 11;26(8):2195. doi: 10.3390/molecules26082195. Molecules. 2021. PMID: 33920352 Free PMC article.
-
Therapeutic potentials of plant iridoids in Alzheimer's and Parkinson's diseases: A review.Eur J Med Chem. 2019 May 1;169:185-199. doi: 10.1016/j.ejmech.2019.03.009. Epub 2019 Mar 8. Eur J Med Chem. 2019. PMID: 30877973 Review.
-
Caenorhabditis elegans Model for Initial Screening and Mechanistic Evaluation of Potential New Drugs for Aging and Alzheimer’s Disease.In: Buccafusco JJ, editor. Methods of Behavior Analysis in Neuroscience. 2nd edition. Boca Raton (FL): CRC Press/Taylor & Francis; 2009. Chapter 16. In: Buccafusco JJ, editor. Methods of Behavior Analysis in Neuroscience. 2nd edition. Boca Raton (FL): CRC Press/Taylor & Francis; 2009. Chapter 16. PMID: 21204333 Free Books & Documents. Review.
Cited by
-
Neuroprotective Role of Phytoconstituents-based Nanoemulsion for the Treatment of Alzheimer's Disease.Curr Top Med Chem. 2024;24(19):1683-1698. doi: 10.2174/0115680266296001240327090111. Curr Top Med Chem. 2024. PMID: 38676489 Review.
-
Characterization and Expression of TGF-β Proteins and Receptor in Sea Cucumber (Holothuria scabra): Insights into Potential Applications via Molecular Docking Predictions.Int J Mol Sci. 2025 Jul 21;26(14):6998. doi: 10.3390/ijms26146998. Int J Mol Sci. 2025. PMID: 40725247 Free PMC article.
-
Plant-Based Inhibitors of Protein Aggregation.Biomolecules. 2025 Mar 25;15(4):481. doi: 10.3390/biom15040481. Biomolecules. 2025. PMID: 40305223 Free PMC article. Review.
-
Olfactory dysfunction as an early pathogenic indicator in C. elegans models of Alzheimer's and polyglutamine diseases.Front Aging Neurosci. 2024 Oct 1;16:1462238. doi: 10.3389/fnagi.2024.1462238. eCollection 2024. Front Aging Neurosci. 2024. PMID: 39411283 Free PMC article.
-
Nicotine-mediated therapy for Parkinson's disease in transgenic Caenorhabditis elegans model.Front Aging Neurosci. 2024 May 15;16:1358141. doi: 10.3389/fnagi.2024.1358141. eCollection 2024. Front Aging Neurosci. 2024. PMID: 38813528 Free PMC article.
References
-
- Leiteritz A., Schmiedl T., Baumanns S., Wenzel U. Amyloid-beta induced paralysis is reduced by cholecalciferol through inhibition of the steroid-signaling pathway in an Alzheimer model of Caenorhabditis elegans. Nutr Neurosci. 2019:1–8. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Other Literature Sources
Miscellaneous
