Nicotinamide as potential biomarker for Alzheimer's disease: A translational study based on metabolomics
- PMID: 36685284
- PMCID: PMC9853457
- DOI: 10.3389/fmolb.2022.1067296
Nicotinamide as potential biomarker for Alzheimer's disease: A translational study based on metabolomics
Abstract
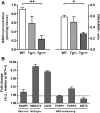
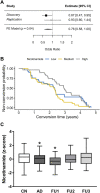
Introduction: The metabolic routes altered in Alzheimer's disease (AD) brain are poorly understood. As the metabolic pathways are evolutionarily conserved, the metabolic profiles carried out in animal models of AD could be directly translated into human studies. Methods: We performed untargeted Nuclear Magnetic Resonance metabolomics in hippocampus of McGill-R-Thy1-APP transgenic (Tg) rats, a model of AD-like cerebral amyloidosis and the translational potential of these findings was assessed by targeted Gas Chromatography-Electron Impact-Mass Spectrometry in plasma of participants in the German longitudinal cohort AgeCoDe. Results: In rat hippocampus 26 metabolites were identified. Of these 26 metabolites, nine showed differences between rat genotypes that were nominally significant. Two of them presented partial least square-discriminant analysis (PLS-DA) loadings with the larger absolute weights and the highest Variable Importance in Projection (VIP) scores and were specifically assigned to nicotinamide adenine dinucleotide (NAD) and nicotinamide (Nam). NAD levels were significantly decreased in Tg rat brains as compared to controls. In agreement with these results, plasma of AD patients showed significantly reduced levels of Nam in respect to cognitively normal participants. In addition, high plasma levels of Nam showed a 27% risk reduction of progressing to AD dementia within the following 2.5 years, this hazard ratio is lost afterwards. Discussion: To our knowledge, this is the first report showing that a decrease of Nam plasma levels is observed couple of years before conversion to AD, thereby suggesting its potential use as biomarker for AD progression.
Keywords: NAD salvage pathway; alzheimer’s disease; biomarkers, brain alterations; case-control analysis; nicotinamide (NAM); transgenic rats; vit B3.
Copyright © 2023 Dalmasso, Arán, Galeano, Perin, Giavalisco, Martino Adami, Novack, Castaño, Cuello, Scherer, Maier, Wagner, Riedel-Heller, Ramirez and Morelli.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
-
- Adams J. D. (2021). Probable causes of Alzheimer’s disease. Sci. 3, 16. 10.3390/sci3010016 - DOI
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

