Identification of kinases activated by multiple pro-angiogenic growth factors
- PMID: 36686695
- PMCID: PMC9847502
- DOI: 10.3389/fphar.2022.1022722
Identification of kinases activated by multiple pro-angiogenic growth factors
Abstract
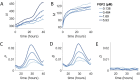
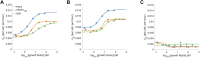
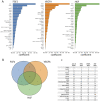
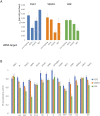
Antiangiogenic therapy began as an effort to inhibit VEGF signaling, which was thought to be the sole factor driving tumor angiogenesis. It has become clear that there are more pro-angiogenic growth factors that can substitute for VEGF during tumor vascularization. This has led to the development of multi-kinase inhibitors which simultaneously target multiple growth factor receptors. These inhibitors perform better than monotherapies yet to date no multi-kinase inhibitor targets all receptors known to be involved in pro-angiogenic signaling and resistance inevitably occurs. Given the large number of pro-angiogenic growth factors identified, it may be impossible to simultaneously target all pro-angiogenic growth factor receptors. Here we search for kinase targets, some which may be intracellularly localized, that are critical in endothelial cell proliferation irrespective of the growth factor used. We develop a quantitative endothelial cell proliferation assay and combine it with "kinome regression" or KIR, a recently developed method capable of identifying kinases that influence a quantitative phenotype. We report the kinases implicated by KIR and provide orthogonal evidence of their importance in endothelial cell proliferation. Our approach may point to a new strategy to develop a more complete anti-angiogenic blockade.
Keywords: cell growth; kinase inhibitors; kinome regularization; polypharmacology; proangiogenic growth factors; tumor vascularization.
Copyright © 2023 Gruver, Rata, Peshkin and Kirschner.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Resistance Mechanisms to Anti-angiogenic Therapies in Cancer.Front Oncol. 2020 Feb 27;10:221. doi: 10.3389/fonc.2020.00221. eCollection 2020. Front Oncol. 2020. PMID: 32175278 Free PMC article. Review.
-
Targeting Angiogenesis in Cancer Therapy: Moving Beyond Vascular Endothelial Growth Factor.Oncologist. 2015 Jun;20(6):660-73. doi: 10.1634/theoncologist.2014-0465. Epub 2015 May 22. Oncologist. 2015. PMID: 26001391 Free PMC article. Review.
-
Salvianolic acid B and ferulic acid synergistically promote angiogenesis in HUVECs and zebrafish via regulating VEGF signaling.J Ethnopharmacol. 2022 Jan 30;283:114667. doi: 10.1016/j.jep.2021.114667. Epub 2021 Sep 29. J Ethnopharmacol. 2022. PMID: 34597652
-
Targeting tumor micro-environment for design and development of novel anti-angiogenic agents arresting tumor growth.Prog Biophys Mol Biol. 2013 Nov;113(2):333-54. doi: 10.1016/j.pbiomolbio.2013.10.001. Epub 2013 Oct 15. Prog Biophys Mol Biol. 2013. PMID: 24139944 Review.
-
Antiangiogenic mechanisms of PJ-8, a novel inhibitor of vascular endothelial growth factor receptor signaling.Carcinogenesis. 2012 May;33(5):1022-30. doi: 10.1093/carcin/bgs127. Epub 2012 Mar 20. Carcinogenesis. 2012. PMID: 22436611
References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

