A pan-cancer landscape of IGF2BPs and their association with prognosis, stemness and tumor immune microenvironment
- PMID: 36686749
- PMCID: PMC9846525
- DOI: 10.3389/fonc.2022.1049183
A pan-cancer landscape of IGF2BPs and their association with prognosis, stemness and tumor immune microenvironment
Abstract
Background: The human insulin-like growth factor 2 mRNA binding proteins 1-3 (IGF2BP1-3, also called IMP1-3) play essential roles in mRNA regulation, including its splicing, translocation, stability, and translation. However, knowledge regarding the involvement of IGF2BPs in tumor immunity and stemness across cancer types is still lacking.
Methods: In this study, we comprehensively analyzed pan-cancer multi-omic data to determine the correlation of IGF2BPs mRNA and protein expression with various cancer parameters such as mutation frequency, prognostic value, the tumor microenvironment (TME), checkpoint blockade, tumor immune infiltration, stemness and drug sensitivity. Validation of the expression of IGF2BPs in cancer samples and glioma cells were performed by quantitative real-time (qRT)-PCR, and immunofluorescence staining. Investigation of the functional role of IGF2BP3 in glioma stem cells(GSCs) were performed by sphere formation, cytotoxicity, transwell, and wound healing assays.
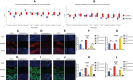
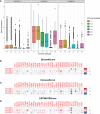
Results: We found that IGF2BP1 and 3 are either absent or expressed at very low levels in most normal tissues. However, IGF2BP1-3 can be re-expressed in a broad range of cancer types and diverse cancer cell lines, where their expression often correlates with poor prognosis. Immunofluorescence staining and qRT-PCR analyses also showed that the expression of IGF2BP2 and IGF2BP3 were higher in cancer tissues than that in adjacent normal tissues. Moreover, IGF2BPs are associated with TME and stemness in human pan-cancer. Remarkably, IGF2BP3 participated in the maintenance and self-renewal of glioma stem cell (GSCs). Knockdown of IGF2BP3 attenuated GSC and glioma cell proliferation, invasion, and migration.
Conclusions: Our systematic pan-cancer study confirmed the identification of IGF2BPs as therapeutic targets and highlighted the need to study their association with stemness, and the TME, which contribute to the cancer drug-discovery research. Especially, preliminary studies demonstrate the IGF2BP3 as a potential negative regulator of glioma tumorigenesis by modulating stemness.
Keywords: glioma stem cells; insulin-like growth factor 2(IGF2) messenger RNA; pan-cancer; prognosis signature; tumor microenvironment.
Copyright © 2023 Shao, Zhao, Zhang, Ding, Guo, Hou, Kan, Deng and Xu.
Conflict of interest statement
Author HZ was employed by Zhengzhou Revogene Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures








Similar articles
-
The role of APOBEC3C in modulating the tumor microenvironment and stemness properties of glioma: evidence from pancancer analysis.Front Immunol. 2023 Sep 21;14:1242972. doi: 10.3389/fimmu.2023.1242972. eCollection 2023. Front Immunol. 2023. PMID: 37809064 Free PMC article.
-
Multi-omics analysis of N6-methyladenosine reader IGF2BP3 as a promising biomarker in pan-cancer.Front Immunol. 2023 Jan 25;14:1071675. doi: 10.3389/fimmu.2023.1071675. eCollection 2023. Front Immunol. 2023. PMID: 36761737 Free PMC article.
-
Insulin-like growth factor 2 mRNA-binding proteins (IGF2BPs): post-transcriptional drivers of cancer progression?Cell Mol Life Sci. 2013 Aug;70(15):2657-75. doi: 10.1007/s00018-012-1186-z. Epub 2012 Oct 16. Cell Mol Life Sci. 2013. PMID: 23069990 Free PMC article. Review.
-
IGF2BP family of RNA-binding proteins regulate innate and adaptive immune responses in cancer cells and tumor microenvironment.Front Immunol. 2023 Jul 12;14:1224516. doi: 10.3389/fimmu.2023.1224516. eCollection 2023. Front Immunol. 2023. PMID: 37503349 Free PMC article.
-
Oncofetal protein IGF2BPs in human cancer: functions, mechanisms and therapeutic potential.Biomark Res. 2023 Jun 6;11(1):62. doi: 10.1186/s40364-023-00499-0. Biomark Res. 2023. PMID: 37280679 Free PMC article. Review.
Cited by
-
High expression of ARPC1B correlates with immune infiltration and poor outcomes in glioblastoma.Biochem Biophys Rep. 2023 Dec 19;37:101619. doi: 10.1016/j.bbrep.2023.101619. eCollection 2024 Mar. Biochem Biophys Rep. 2023. PMID: 38188361 Free PMC article.
-
Deciphering the Divergent Gene Expression Landscapes of m6A/m5C/m1A Methylation Regulators in Hepatocellular Carcinoma Through Single-Cell and Bulk RNA Transcriptomic Analysis.J Hepatocell Carcinoma. 2023 Dec 28;10:2383-2395. doi: 10.2147/JHC.S448047. eCollection 2023. J Hepatocell Carcinoma. 2023. PMID: 38164510 Free PMC article.
-
Low-density lipoprotein receptor promotes crosstalk between cell stemness and tumor immune microenvironment in breast cancer: a large data-based multi-omics study.J Transl Med. 2023 Nov 30;21(1):871. doi: 10.1186/s12967-023-04699-y. J Transl Med. 2023. PMID: 38037058 Free PMC article.
-
Three Liquid-Liquid Phase Separation-Related Genes Associated with Prognosis in Glioma.Pharmgenomics Pers Med. 2024 Apr 22;17:171-181. doi: 10.2147/PGPM.S442000. eCollection 2024. Pharmgenomics Pers Med. 2024. PMID: 38681062 Free PMC article.
-
Old players and new insights: unraveling the role of RNA-binding proteins in brain tumors.Theranostics. 2025 Apr 13;15(11):5238-5257. doi: 10.7150/thno.113312. eCollection 2025. Theranostics. 2025. PMID: 40303323 Free PMC article. Review.
References
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

