New insight into strategies used to develop long-acting G-CSF biologics for neutropenia therapy
- PMID: 36686781
- PMCID: PMC9850083
- DOI: 10.3389/fonc.2022.1026377
New insight into strategies used to develop long-acting G-CSF biologics for neutropenia therapy
Abstract
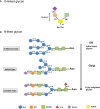
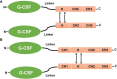
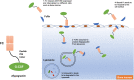
Over the last 20 years, granulocyte colony-stimulating factors (G-CSFs) have become the major therapeutic option for the treatment of patients with neutropenia. Most of the current G-CSFs require daily injections, which are inconvenient and expensive for patients. Increased understanding of G-CSFs' structure, expression, and mechanism of clearance has been very instrumental in the development of new generations of long-acting G-CSFs with improved efficacy. Several approaches to reducing G-CSF clearance via conjugation techniques have been investigated. PEGylation, glycosylation, polysialylation, or conjugation with immunoglobulins or albumins have successfully increased G-CSFs' half-lives. Pegfilgrastim (Neulasta) has been successfully approved and marketed for the treatment of patients with neutropenia. The rapidly expanding market for G-CSFs has increased demand for G-CSF biosimilars. Therefore, the importance of this review is to highlight the principle, elimination's route, half-life, clearance, safety, benefits, and limitations of different strategies and techniques used to increase the half-life of biotherapeutic G-CSFs. Understanding these strategies will allow for a new treatment with more competitive manufacturing and lower unit costs compared with that of Neulasta.
Keywords: G-CSF; long-acting; neutropenia; strategy; therapy.
Copyright © 2023 Theyab, Alsharif, Alzahrani, Oyouni, Hawsawi, Algahtani, Alghamdi and Alshammary.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures



Similar articles
-
New G-CSF agonists for neutropenia therapy.Expert Opin Investig Drugs. 2014 Jan;23(1):21-35. doi: 10.1517/13543784.2013.838558. Epub 2013 Sep 27. Expert Opin Investig Drugs. 2014. PMID: 24073859 Review.
-
2010 update of EORTC guidelines for the use of granulocyte-colony stimulating factor to reduce the incidence of chemotherapy-induced febrile neutropenia in adult patients with lymphoproliferative disorders and solid tumours.Eur J Cancer. 2011 Jan;47(1):8-32. doi: 10.1016/j.ejca.2010.10.013. Epub 2010 Nov 20. Eur J Cancer. 2011. PMID: 21095116
-
Systematic Review and Meta-analysis of Short- versus Long-Acting Granulocyte Colony-Stimulating Factors for Reduction of Chemotherapy-Induced Febrile Neutropenia.Adv Ther. 2018 Nov;35(11):1816-1829. doi: 10.1007/s12325-018-0798-6. Epub 2018 Oct 8. Adv Ther. 2018. PMID: 30298233 Free PMC article.
-
Pegfilgrastim for the prevention of chemotherapy-induced febrile neutropenia in patients with solid tumors.Expert Opin Biol Ther. 2015;15(12):1799-817. doi: 10.1517/14712598.2015.1101063. Epub 2015 Oct 21. Expert Opin Biol Ther. 2015. PMID: 26488491 Review.
-
Cost-effectiveness of granulocyte colony-stimulating factor prophylaxis for febrile neutropenia in breast cancer in the United Kingdom.Value Health. 2011 Jun;14(4):465-74. doi: 10.1016/j.jval.2010.10.037. Epub 2011 Apr 22. Value Health. 2011. PMID: 21669371
Cited by
-
Delivery of DNA-Based Therapeutics for Treatment of Chronic Diseases.Pharmaceutics. 2024 Apr 13;16(4):535. doi: 10.3390/pharmaceutics16040535. Pharmaceutics. 2024. PMID: 38675196 Free PMC article. Review.
-
Flavopiridol: a promising cyclin-dependent kinase inhibitor in cancer treatment.Naunyn Schmiedebergs Arch Pharmacol. 2025 Apr;398(4):3489-3511. doi: 10.1007/s00210-024-03599-2. Epub 2024 Nov 26. Naunyn Schmiedebergs Arch Pharmacol. 2025. PMID: 39589530 Review.
-
Implications of glycosylation for the development of selected cytokines and their derivatives for medical use.Biotechnol Adv. 2024 Dec;77:108467. doi: 10.1016/j.biotechadv.2024.108467. Epub 2024 Oct 22. Biotechnol Adv. 2024. PMID: 39447666 Free PMC article. Review.
-
Population Pharmacokinetic-Pharmacodynamic Modeling of Granulocyte Colony-Stimulating Factor to Optimize Dosing and Timing for CD34+ Cell Harvesting.Clin Transl Sci. 2025 Jan;18(1):e70121. doi: 10.1111/cts.70121. Clin Transl Sci. 2025. PMID: 39748466 Free PMC article.
-
Granulocyte Colony Stimulating Factor-Mobilized Peripheral Blood Mononuclear Cells: An Alternative Cellular Source for Chimeric Antigen Receptor Therapy.Int J Mol Sci. 2024 May 25;25(11):5769. doi: 10.3390/ijms25115769. Int J Mol Sci. 2024. PMID: 38891957 Free PMC article. Review.
References
-
- Inc. A. Neulasta (pegfilgrastim) prescribing information. Initial U.S. Approval. (California: Amgen Inc. One Amgen Center Drive Thousand Oaks; ) (2002) 91320-1799 U.S. License No. 1080.
Publication types
LinkOut - more resources
Full Text Sources

