Silver-catalyzed direct selanylation of indoles: synthesis and mechanistic insights
- PMID: 36686957
- PMCID: PMC9811358
- DOI: 10.1039/d2ra06813c
Silver-catalyzed direct selanylation of indoles: synthesis and mechanistic insights
Abstract
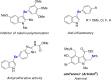
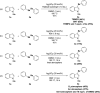
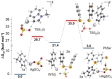
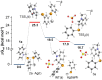
Herein we describe the Ag(i)-catalyzed direct selanylation of indoles with diorganoyl diselenides. The reaction gave 3-selanylindoles with high regioselectivity and also allowed direct access to 2-selanylindoles when the C3 position of the indole ring was blocked via a process similar to Plancher rearrangement. Experimental analyses and density functional theory calculations were carried out in order to picture the reaction mechanism. Among the pathways considered (via concerted metalation-deprotonation, Ag(iii), radical, and electrophilic aromatic substitution), our findings support a classic electrophilic aromatic substitution via Lewis adducts between Ag(i) and diorganoyl diselenides. The results also afforded new insights into the interactions between Ag(i) and diorganoyl diselenides.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures
Similar articles
-
Bismuth(III)-Catalyzed Regioselective Selenation of Indoles with Diaryl Diselenides: Synthesis of 3-Selanylindoles.Molecules. 2024 Jul 8;29(13):3227. doi: 10.3390/molecules29133227. Molecules. 2024. PMID: 38999179 Free PMC article.
-
Sonochemistry: An efficient alternative to the synthesis of 3-selanylindoles using CuI as catalyst.Ultrason Sonochem. 2015 Nov;27:192-199. doi: 10.1016/j.ultsonch.2015.05.012. Epub 2015 May 22. Ultrason Sonochem. 2015. PMID: 26186837
-
Cooperative Au/Ag Dual-Catalyzed Cross-Dehydrogenative Biaryl Coupling: Reaction Development and Mechanistic Insight.J Am Chem Soc. 2019 Feb 20;141(7):3187-3197. doi: 10.1021/jacs.8b12929. Epub 2019 Feb 8. J Am Chem Soc. 2019. PMID: 30681846
-
Mechanism of Rhodium-Catalyzed C-H Functionalization: Advances in Theoretical Investigation.Acc Chem Res. 2017 Nov 21;50(11):2799-2808. doi: 10.1021/acs.accounts.7b00400. Epub 2017 Nov 7. Acc Chem Res. 2017. PMID: 29112396
-
Diorganyl diselenides: a powerful tool for the construction of selenium containing scaffolds.Dalton Trans. 2021 Sep 28;50(37):12764-12790. doi: 10.1039/d1dt01982a. Dalton Trans. 2021. PMID: 34581339 Review.
Cited by
-
Bismuth(III)-Catalyzed Regioselective Selenation of Indoles with Diaryl Diselenides: Synthesis of 3-Selanylindoles.Molecules. 2024 Jul 8;29(13):3227. doi: 10.3390/molecules29133227. Molecules. 2024. PMID: 38999179 Free PMC article.
-
Copper(II)-Catalyzed Direct C3 Chalcogenylation of Indoles.Molecules. 2025 Apr 22;30(9):1870. doi: 10.3390/molecules30091870. Molecules. 2025. PMID: 40363677 Free PMC article.
-
Selenium Modification of Natural Products and Its Research Progress.Foods. 2023 Oct 13;12(20):3773. doi: 10.3390/foods12203773. Foods. 2023. PMID: 37893666 Free PMC article. Review.
-
Synthesis of Selenium-Decorated N-Oxide Isoquinolines: Arylseleninic Acids in Selenocyclization Reactions.J Org Chem. 2024 Aug 16;89(16):11272-11280. doi: 10.1021/acs.joc.4c00944. Epub 2024 Aug 1. J Org Chem. 2024. PMID: 39088563 Free PMC article.
References
-
- Li C.-J. and Bi X., Silver Catalysis in Organic Synthesis, Wiley, 1st edn, 2019, vol. 2
- Hermata M., Silver in Organic Chemistry, Wiley, 1st edn, 2011
- Echavarren A. M. Jiao N. Gevorgyan V. Coinage metals in organic synthesis. Chem. Soc. Rev. 2016;45:4445. - PubMed
- Jordan A. J. Lalic G. Sadighi J. P. Coinage metal hydrides: synthesis, characterization, and reactivity. Chem. Rev. 2016;116:8318. - PubMed
- Lo V. K.-Y. Chan A. O.-Y. Che C.-M. Gold and silver catalysis: from organic transformation to bioconjugation. Org. Biomol. Chem. 2015;13:6667. - PubMed
- Díaz-Requejo M. M. Pérez P. J. Coinage metal catalyzed C-H bond functionalization of hydrocarbons. Chem. Rev. 2008;108:3379. - PubMed
- Lipshutz B. H. Yamamoto Y. Introduction: coinage metals in organic synthesis. Chem. Rev. 2008;108:2793. - PubMed
-
- Fang G. Bi X. Silver-catalysed reactions of alkynes: recent advances. Chem. Soc. Rev. 2015;44:8124. - PubMed
- Pelissier H. Enantioselective silver-catalyzed transformations. Chem. Rev. 2016;116:14868. - PubMed
- Radhika S. Abdulla C. M. A. Aneeja T. Anilkumar G. Silver-catalysed C-H bond activation: a recent review. New J. Chem. 2021;45:15718.
- Sekine K. Yamada T. Sylver-catalyzed carboxylation. Chem. Soc. Rev. 2016;45:4524. - PubMed
- Naodovic M. Yamamoto H. Asymmetric silver-catalyzed reactions. Chem. Rev. 2008;108:3132. - PubMed
- Kaur N. Ahlawat N. Bhardwaj P. Verma Y. Grewal P. Jangid N. K. Ag-mediated synthesis of six-membered N-heterocycles. Synth. Commun. 2020;50:753.
- Neetha M. Aneeja T. Afsina C. M. A. Anilkumar G. An Overview of Ag-catalyzed Synthesis of Six-membered Heterocycles. ChemCatChem. 2020;12:5330.
- Li P. Wu W. Jiang H. Recent Advances in Silver-Catalyzed Transformations of Electronically Unbiased Alkenes and Alkynes. ChemCatChem. 2020;12:5034.
- Álvarez-Corral M. Muñoz-Dorado M. Rodríguez-García I. Silver-mediated synthesis of heterocycles. Chem. Rev. 2008;108:3174. - PubMed
- Sivaguru P. Cao S. Babu K. R. Bi X. Silver-catalyzed activation of terminal alkynes for synthesizing nitrogen-containing molecules. Acc. Chem. Res. 2020;53:662. - PubMed
-
- Wen C. Yin A. Dai W.-L. Recent advances in silver-based heterogeneous catalysts for green chemistry process. Appl. Catal., B. 2014;160:730.
- Millar G. J. Collins M. Industrial production of formaldehyde using polycrystalline silver catalyst. Ind. Eng. Chem. Res. 2017;56:9247.
-
- Gorin D. J. Toste F. D. Relativistic effects in homogeneous gold catalysis. Nature. 2007;446:395. - PubMed
- Pyykkö P. Theoretical chemistry of gold. Angew. Chem., Int. Ed. 2004;43:4412. - PubMed
- Bagus P. S. Lee Y. S. Pitzer K. S. Effects of relativity and of the lanthanide contraction on the atoms from hafnium to bismuth. Chem. Phys. Lett. 1975;33:408.
-
-
Selected examples:
- Jia F. Zhang B. Mechanistic insight into the silver-catalyzed cycloaddition synthesis of 1,4-disubstituted-1,2,3-triazoles: the key of the role of silver. New J. Chem. 2019;43:8634.
- Wang S. Yang L.-J. Zeng J.-L. Zheng Y. Ma J.-A. Silver-catalyzed [3+2] cycloaddition of isocyanides with diazo compounds: new regioselective access to 1,4-disubstituted-1,2,3-triazoles. Org. Chem. Front. 2015;2:1468.
- McNulty J. Keskar K. Vemula R. The First Well-Defined Silver (I)-Complex-Catalyzed Cycloaddition of Azides onto Terminal Alkynes at Room Temperature. Chem.–Eur. J. 2011;17:14727. - PubMed
- Nakamura I. Nemoto T. Yamamoto Y. Meijere A. Thermally induced and silver-salt-catalyzed [2+2] cycloadditions of imines to (alkoxymethylene)cyclopropanes. Angew. Chem., Int. Ed. 2006;45:5176. - PubMed
- Zhu S. Liang R. Jiang H. Wu W. An Efficient Route to Polysubstituted Tetrahydronaphtols: Silver-Catalyzed [4+2] Cyclization of 2-Alkylbenzaldehydes and Alkene. Angew. Chem., Int. Ed. 2012;51:10861. - PubMed
- Gao M. Chen H. Bai R. Cheng B. Lei A. Synthesis of pyrroles by click reaction: silver-catalyzed cycloaddition of terminal alkynes with isocyanides. Angew. Chem., Int. Ed. 2013;52:6958. - PubMed
-
LinkOut - more resources
Full Text Sources
Miscellaneous

