Neurotransmitter content heterogeneity within an interneuron class shapes inhibitory transmission at a central synapse
- PMID: 36687523
- PMCID: PMC9846633
- DOI: 10.3389/fncel.2022.1060189
Neurotransmitter content heterogeneity within an interneuron class shapes inhibitory transmission at a central synapse
Abstract
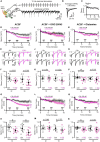
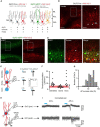
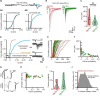
Neurotransmitter content is deemed the most basic defining criterion for neuronal classes, contrasting with the intercellular heterogeneity of many other molecular and functional features. Here we show, in the adult mouse brain, that neurotransmitter content variegation within a neuronal class is a component of its functional heterogeneity. Golgi cells (GoCs), the well-defined class of cerebellar interneurons inhibiting granule cells (GrCs), contain cytosolic glycine, accumulated by the neuronal transporter GlyT2, and GABA in various proportions. By performing acute manipulations of cytosolic GABA and glycine supply, we find that competition of glycine with GABA reduces the charge of IPSC evoked in GrCs and, more specifically, the amplitude of a slow component of the IPSC decay. We then pair GrCs recordings with optogenetic stimulations of single GoCs, which preserve the intracellular transmitter mixed content. We show that the strength and decay kinetics of GrCs IPSCs, which are entirely mediated by GABAA receptors, are negatively correlated to the presynaptic expression of GlyT2 by GoCs. We isolate a slow spillover component of GrCs inhibition that is also affected by the expression of GlyT2, leading to a 56% decrease in relative charge. Our results support the hypothesis that presynaptic loading of glycine negatively impacts the GABAergic transmission in mixed interneurons, most likely through a competition for vesicular filling. We discuss how the heterogeneity of neurotransmitter supply within mixed interneurons like the GoC class may provide a presynaptic mechanism to tune the gain of microcircuits such as the granular layer, thereby expanding the realm of their possible dynamic behaviors.
Keywords: cerebellum; co-transmission; golgi cells; granule cells; inhibition.
Copyright © 2023 Dumontier, Mailhes-Hamon, Supplisson and Dieudonné.
Conflict of interest statement
SD is a stakeholder of the SME Karthala System and an inventor of IP licensed to this company. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
Pathway-Specific Drive of Cerebellar Golgi Cells Reveals Integrative Rules of Cortical Inhibition.J Neurosci. 2019 Feb 13;39(7):1169-1181. doi: 10.1523/JNEUROSCI.1448-18.2018. Epub 2018 Dec 26. J Neurosci. 2019. PMID: 30587539 Free PMC article.
-
Co-localization of glycine and gaba immunoreactivity in interneurons in Macaca monkey cerebellar cortex.Neuroscience. 2006 Sep 15;141(4):1951-9. doi: 10.1016/j.neuroscience.2006.05.012. Epub 2006 Jun 19. Neuroscience. 2006. PMID: 16784818
-
Active Dendrites and Differential Distribution of Calcium Channels Enable Functional Compartmentalization of Golgi Cells.J Neurosci. 2015 Nov 25;35(47):15492-504. doi: 10.1523/JNEUROSCI.3132-15.2015. J Neurosci. 2015. PMID: 26609148 Free PMC article.
-
Rapid, activity-independent turnover of vesicular transmitter content at a mixed glycine/GABA synapse.J Neurosci. 2013 Mar 13;33(11):4768-81. doi: 10.1523/JNEUROSCI.5555-12.2013. J Neurosci. 2013. PMID: 23486948 Free PMC article.
-
Presynaptic control of inhibitory neurotransmitter content in VIAAT containing synaptic vesicles.Neurochem Int. 2016 Sep;98:94-102. doi: 10.1016/j.neuint.2016.06.002. Epub 2016 Jun 11. Neurochem Int. 2016. PMID: 27296116 Review.
Cited by
-
Interactions Involving Glycine and Other Amino Acid Neurotransmitters: Focus on Transporter-Mediated Regulation of Release and Glycine-Glutamate Crosstalk.Biomedicines. 2024 Jul 8;12(7):1518. doi: 10.3390/biomedicines12071518. Biomedicines. 2024. PMID: 39062091 Free PMC article. Review.
-
Revisiting the development of cerebellar inhibitory interneurons in the light of single-cell genetic analyses.Histochem Cell Biol. 2024 Jan;161(1):5-27. doi: 10.1007/s00418-023-02251-z. Epub 2023 Nov 8. Histochem Cell Biol. 2024. PMID: 37940705 Free PMC article. Review.
-
A Gene-Expression Based Comparison of Murine and Human Inhibitory Interneurons in the Cerebellar Cortex and Nuclei.Cerebellum. 2025 Feb 28;24(2):55. doi: 10.1007/s12311-025-01809-y. Cerebellum. 2025. PMID: 40019676 Free PMC article.
-
Coincidence detection between apical and basal dendrites drives STDP in cerebellar Golgi cells.Commun Biol. 2025 May 12;8(1):731. doi: 10.1038/s42003-025-08153-1. Commun Biol. 2025. PMID: 40350534 Free PMC article.
-
Variations on the theme: focus on cerebellum and emotional processing.Front Syst Neurosci. 2023 May 10;17:1185752. doi: 10.3389/fnsys.2023.1185752. eCollection 2023. Front Syst Neurosci. 2023. PMID: 37234065 Free PMC article. Review.
References
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources

