Phage-encoded carbohydrate-interacting proteins in the human gut
- PMID: 36687636
- PMCID: PMC9853417
- DOI: 10.3389/fmicb.2022.1083208
Phage-encoded carbohydrate-interacting proteins in the human gut
Abstract
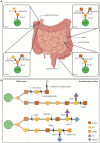
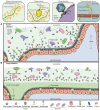
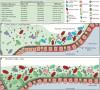
In the human gastrointestinal tract, the gut mucosa and the bacterial component of the microbiota interact and modulate each other to accomplish a variety of critical functions. These include digestion aid, maintenance of the mucosal barrier, immune regulation, and production of vitamins, hormones, and other metabolites that are important for our health. The mucus lining of the gut is primarily composed of mucins, large glycosylated proteins with glycosylation patterns that vary depending on factors including location in the digestive tract and the local microbial population. Many gut bacteria have evolved to reside within the mucus layer and thus encode mucus-adhering and -degrading proteins. By doing so, they can influence the integrity of the mucus barrier and therefore promote either health maintenance or the onset and progression of some diseases. The viral members of the gut - mostly composed of bacteriophages - have also been shown to have mucus-interacting capabilities, but their mechanisms and effects remain largely unexplored. In this review, we discuss the role of bacteriophages in influencing mucosal integrity, indirectly via interactions with other members of the gut microbiota, or directly with the gut mucus via phage-encoded carbohydrate-interacting proteins. We additionally discuss how these phage-mucus interactions may influence health and disease states.
Keywords: bacteriophage; glycans; glycosylation; gut; mucins; mucus; mucus-binding; mucus-degrading.
Copyright © 2023 Rothschild-Rodriguez, Hedges, Kaplan, Karav and Nobrega.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Mucus barrier, mucins and gut microbiota: the expected slimy partners?Gut. 2020 Dec;69(12):2232-2243. doi: 10.1136/gutjnl-2020-322260. Epub 2020 Sep 11. Gut. 2020. PMID: 32917747 Free PMC article. Review.
-
Bacteriophages evolve enhanced persistence to a mucosal surface.Proc Natl Acad Sci U S A. 2022 Jul 5;119(27):e2116197119. doi: 10.1073/pnas.2116197119. Epub 2022 Jun 29. Proc Natl Acad Sci U S A. 2022. PMID: 35767643 Free PMC article.
-
The role of glycosylated mucins in maintaining intestinal homeostasis and gut health.Anim Nutr. 2025 Apr 4;21:439-446. doi: 10.1016/j.aninu.2025.03.004. eCollection 2025 Jun. Anim Nutr. 2025. PMID: 40491555 Free PMC article. Review.
-
The Interaction of the Gut Microbiota with the Mucus Barrier in Health and Disease in Human.Microorganisms. 2018 Aug 2;6(3):78. doi: 10.3390/microorganisms6030078. Microorganisms. 2018. PMID: 30072673 Free PMC article. Review.
-
The glycoconjugate-degrading enzymes of Clostridium perfringens: Tailored catalysts for breaching the intestinal mucus barrier.Glycobiology. 2021 Jun 29;31(6):681-690. doi: 10.1093/glycob/cwaa050. Glycobiology. 2021. PMID: 32472136 Review.
Cited by
-
A compendium of viruses from methanogenic archaea reveals their diversity and adaptations to the gut environment.Nat Microbiol. 2023 Nov;8(11):2170-2182. doi: 10.1038/s41564-023-01485-w. Epub 2023 Sep 25. Nat Microbiol. 2023. PMID: 37749252
-
Antimicrobial resistance: use of phage therapy in the management of resistant infections.Mol Biol Rep. 2024 Aug 21;51(1):925. doi: 10.1007/s11033-024-09870-2. Mol Biol Rep. 2024. PMID: 39167154 Review.
-
The colonic mucosal virome in inflammatory bowel disease reveals Crassvirales depletion and disease-specific virome features.Gut Microbes. 2025 Dec;17(1):2539450. doi: 10.1080/19490976.2025.2539450. Epub 2025 Aug 3. Gut Microbes. 2025. PMID: 40754936 Free PMC article.
-
An extended catalog of integrated prophages in the infant and adult fecal microbiome shows high prevalence of lysogeny.Front Microbiol. 2023 Sep 5;14:1254535. doi: 10.3389/fmicb.2023.1254535. eCollection 2023. Front Microbiol. 2023. PMID: 37731926 Free PMC article.
-
Mucin and Agitation Shape Predation of Escherichia coli by Lytic Coliphage.Microorganisms. 2023 Feb 17;11(2):508. doi: 10.3390/microorganisms11020508. Microorganisms. 2023. PMID: 36838472 Free PMC article.
References
Publication types
LinkOut - more resources
Full Text Sources

