Computational insight into a mechanistic overview of water exchange kinetics and thermodynamic stabilities of bis and tris-aquated complexes of lanthanides
- PMID: 36688060
- PMCID: PMC9816859
- DOI: 10.1039/d2ra05810c
Computational insight into a mechanistic overview of water exchange kinetics and thermodynamic stabilities of bis and tris-aquated complexes of lanthanides
Abstract
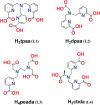
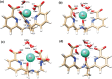
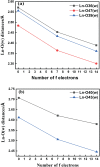
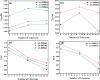
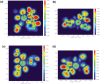
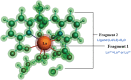
A thorough investigation of Ln3+ complexes with more than one inner-sphere water molecule is crucial for designing high relaxivity contrast agents (CAs) used in magnetic resonance imaging (MRI). This study accomplished a comparative stability analysis of two hexadentate (H3cbda and H3dpaa) and two heptadentate (H4peada and H3tpaa) ligands with Ln3+ ions. The higher stability of the hexadentate H3cbda and heptadentate H4peada ligands has been confirmed by the binding affinity and Gibbs free energy analysis in aqueous solution. In addition, energy decomposition analysis (EDA) reveals the higher binding affinity of the peada4- ligand than the cbda3- ligand towards Ln3+ ions due to the higher charge density of the peada4- ligand. Moreover, a mechanistic overview of water exchange kinetics has been carried out based on the strength of the metal-water bond. The strength of the metal-water bond follows the trend Gd-O47 (w) > Gd-O39 (w) > Gd-O36 (w) in the case of the tris-aquated [Gd(cbda)(H2O)3] and Gd-O43 (w) > Gd-O40 (w) for the bis-aquated [Gd(peada)(H2O)2]- complex, which was confirmed by bond length, electron density (ρ), and electron localization function (ELF) at the corresponding bond critical points. Our analysis also predicts that the activation energy barrier decreases with the decrease in bond strength; hence k ex increases. The 17O and 1H hyperfine coupling constant values of all the coordinated water molecules were different, calculated by using the second-order Douglas-Kroll-Hess (DKH2) approach. Furthermore, the ionic nature of the bonding in the metal-ligand (M-L) bond was confirmed by the Quantum Theory of Atoms-In-Molecules (QTAIM) and ELF along with energy decomposition analysis (EDA). We hope that the results can be used as a basis for the design of highly efficient Gd(iii)-based high relaxivity MRI contrast agents for medical applications.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
There are no conflicts to declare.
Figures
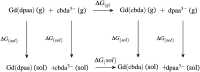




Similar articles
-
Equilibrium and NMR relaxometric studies on the s-triazine-based heptadentate ligand PTDITA showing high selectivity for Gd3+ ions.Inorg Chem. 2012 Feb 20;51(4):2597-607. doi: 10.1021/ic202559h. Epub 2012 Feb 7. Inorg Chem. 2012. PMID: 22313334
-
Unexpected aggregation of neutral, xylene-cored dinuclear GdIII chelates in aqueous solution.Chemistry. 2006 Sep 6;12(26):6841-51. doi: 10.1002/chem.200501335. Chemistry. 2006. PMID: 16770815
-
Stability, water exchange, and anion binding studies on lanthanide(III) complexes with a macrocyclic ligand based on 1,7-diaza-12-crown-4: extremely fast water exchange on the Gd3+ complex.Inorg Chem. 2009 Sep 21;48(18):8878-89. doi: 10.1021/ic9011197. Inorg Chem. 2009. PMID: 19655713
-
Toward the Prediction of Water Exchange Rates in Magnetic Resonance Imaging Contrast Agents: A Density Functional Theory Study.J Phys Chem A. 2015 Jun 18;119(24):6436-45. doi: 10.1021/acs.jpca.5b01728. Epub 2015 Jun 4. J Phys Chem A. 2015. PMID: 26000832
-
Lower denticity leading to higher stability: structural and solution studies of Ln(III)-OBETA complexes.Inorg Chem. 2014 Dec 1;53(23):12499-511. doi: 10.1021/ic5020225. Epub 2014 Nov 11. Inorg Chem. 2014. PMID: 25387307
References
-
- Parker D. Williams J. G. J. Chem. Soc., Dalton Trans. 1996:3613–3628.
-
- Parker D. Dickins R. S. Puschmann H. Crossland C. Howard J. A. Chem. Rev. 2002;102:1977–2010. - PubMed
-
- Cheisson T. Schelter E. J. Science. 2019;363:489–493. - PubMed
-
- Shavaleev N. M. Eliseeva S. V. Scopelliti R. Bünzli J.-C. G. Chem. - Eur. J. 2009;15:10790–10802. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

