Entomopathogenic potential of bacteria associated with soil-borne nematodes and insect immune responses to their infection
- PMID: 36689436
- PMCID: PMC10045567
- DOI: 10.1371/journal.pone.0280675
Entomopathogenic potential of bacteria associated with soil-borne nematodes and insect immune responses to their infection
Abstract
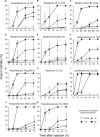
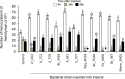
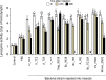
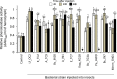
Soil-borne nematodes establish close associations with several bacterial species. Whether they confer benefits to their hosts has been investigated in only a few nematode-bacteria systems. Their ecological function, therefore, remains poorly understood. In this study, we isolated several bacterial species from rhabditid nematodes, molecularly identified them, evaluated their entomopathogenic potential on Galleria mellonella larvae, and measured immune responses of G. mellonella larvae to their infection. Bacteria were isolated from Acrobeloides sp., A. bodenheimeri, Heterorhabditis bacteriophora, Oscheius tipulae, and Pristionchus maupasi nematodes. They were identified as Acinetobacter sp., Alcaligenes sp., Bacillus cereus, Enterobacter sp., Kaistia sp., Lysinibacillus fusiformis, Morganella morganii subsp. morganii, Klebsiella quasipneumoniae subsp. quasipneumoniae, and Pseudomonas aeruginosa. All bacterial strains were found to be highly entomopathogenic as they killed at least 53.33% G. mellonella larvae within 72h post-infection, at a dose of 106 CFU/larvae. Among them, Lysinibacillus fusiformis, Enterobacter sp., Acinetobacter sp., and K. quasipneumoniae subsp. quasipneumoniae were the most entomopathogenic bacteria. Insects strongly responded to bacterial infection. However, their responses were apparently little effective to counteract bacterial infection. Our study, therefore, shows that bacteria associated with soil-borne nematodes have entomopathogenic capacities. From an applied perspective, our study motivates more research to determine the potential of these bacterial strains as biocontrol agents in environmentally friendly and sustainable agriculture.
Copyright: © 2023 Loulou et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
References
-
- Glare TR, Jurat-Fuentes J-L, O’Callaghan M. Basic and Applied, Research: Entomopathogenic Bacteria. Microbial Control of Insect and Mite Pests. 2017; 47–67.
-
- Kalha CS, Singh PP, Kang SS, Hunjan MS, Gupta V, Sharma R. Integrated pest management: Current concepts and ecological perspective. Entomopathogenic viruses and bacteria for insect-pest control. Academic Press. 2014; 225–244.
-
- Sakr HH. Insecticidal activity of Brevibacillus brevis (Paenibacillaceae) bacterial culture filtrate on Spodoptera littoralis (Lepidoptera: Noctuidae) larvae. The Egyptian Society of Experimental Biol. 2017; 13: 331–342.
-
- Gouli VV, Marcelino JAP, Gouli SY. Microbial Pesticides: Biological Resources, Production and Application. Chapter 1 The basic biological resources for the production of microbial pesticides. Elsevier Inc. 2021; 1–134. 10.1016/C2020-0-00269-0. - DOI
Publication types
MeSH terms
Substances
Supplementary concepts
LinkOut - more resources
Full Text Sources
Medical

