A dynamic interplay between chitin synthase and the proteins Expansion/Rebuf reveals that chitin polymerisation and translocation are uncoupled in Drosophila
- PMID: 36689563
- PMCID: PMC9894549
- DOI: 10.1371/journal.pbio.3001978
A dynamic interplay between chitin synthase and the proteins Expansion/Rebuf reveals that chitin polymerisation and translocation are uncoupled in Drosophila
Abstract
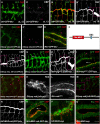
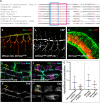
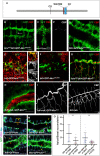
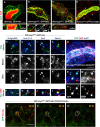
Chitin is a highly abundant polymer in nature and a principal component of apical extracellular matrices in insects. In addition, chitin has proved to be an excellent biomaterial with multiple applications. In spite of its importance, the molecular mechanisms of chitin biosynthesis and chitin structural diversity are not fully elucidated yet. To investigate these issues, we use Drosophila as a model. We previously showed that chitin deposition in ectodermal tissues requires the concomitant activities of the chitin synthase enzyme Kkv and the functionally interchangeable proteins Exp and Reb. Exp/Reb are conserved proteins, but their mechanism of activity during chitin deposition has not been elucidated yet. Here, we carry out a cellular and molecular analysis of chitin deposition, and we show that chitin polymerisation and chitin translocation to the extracellular space are uncoupled. We find that Kkv activity in chitin translocation, but not in polymerisation, requires the activity of Exp/Reb, and in particular of its conserved Nα-MH2 domain. The activity of Kkv in chitin polymerisation and translocation correlate with Kkv subcellular localisation, and in absence of Kkv-mediated extracellular chitin deposition, chitin accumulates intracellularly as membrane-less punctae. Unexpectedly, we find that although Kkv and Exp/Reb display largely complementary patterns at the apical domain, Exp/Reb activity nonetheless regulates the topological distribution of Kkv at the apical membrane. We propose a model in which Exp/Reb regulate the organisation of Kkv complexes at the apical membrane, which, in turn, regulates the function of Kkv in extracellular chitin translocation.
Copyright: © 2023 De Giorgio et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures

References
-
- Zhao X, Zhang J, Zhu KY. Chito-Protein Matrices in Arthropod Exoskeletons and Peritrophic Matrices. In: Cohen E, Merzendorfer H. editors. Extracellular Sugar-Based Biopolymers Matrices Biologically-Inspired Systems. 12. Cham: Springer; 2019.
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Molecular Biology Databases

