New benzothiazole hybrids as potential VEGFR-2 inhibitors: design, synthesis, anticancer evaluation, and in silico study
- PMID: 36691927
- PMCID: PMC9879182
- DOI: 10.1080/14756366.2023.2166036
New benzothiazole hybrids as potential VEGFR-2 inhibitors: design, synthesis, anticancer evaluation, and in silico study
Abstract
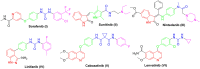
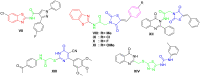
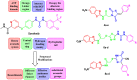
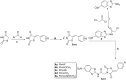
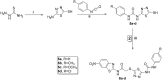
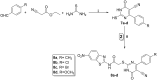
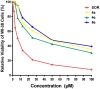
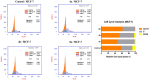
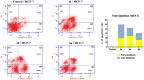
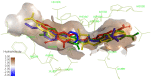
A new series of 2-aminobenzothiazole hybrids linked to thiazolidine-2,4-dione 4a-e, 1,3,4-thiadiazole aryl urea 6a-d, and cyanothiouracil moieties 8a-d was synthesised. The in vitro antitumor effect of the new hybrids was assessed against three cancer cell lines, namely, HCT-116, HEPG-2, and MCF-7 using Sorafenib (SOR) as a standard drug. Among the tested compounds, 4a was the most potent showing IC50 of 5.61, 7.92, and 3.84 µM, respectively. Furthermore, compounds 4e and 8a proved to have strong impact on breast cancer cell line with IC50 of 6.11 and 10.86 µM, respectively. The three compounds showed a good safety profile towards normal WI-38 cells. Flow cytometric analysis of the three compounds in MCF-7 cells revealed that compounds 4a and 4c inhibited cell population in the S phase, whereas 8a inhibited the population in the G1/S phase. The most promising compounds were subjected to a VEGFR-2 inhibitory assay where 4a emerged as the best active inhibitor of VEGFR-2 with IC50 91 nM, compared to 53 nM for SOR. In silico analysis showed that the three new hybrids succeeded to link to the active site like the co-crystallized inhibitor SOR.
Keywords: 134-thiadiazoles; 2-Aminobenzothiazole; VEGFR-2 inhibition; cyanothiouracils; thiazolidine-24-diones.
Conflict of interest statement
The authors report no conflicts of interest.
Figures
Similar articles
-
Design, Synthesis, In Vitro Anti-cancer Activity, ADMET Profile and Molecular Docking of Novel Triazolo[3,4-a]phthalazine Derivatives Targeting VEGFR-2 Enzyme.Anticancer Agents Med Chem. 2018;18(8):1184-1196. doi: 10.2174/1871520618666180412123833. Anticancer Agents Med Chem. 2018. PMID: 29651967
-
Novel benzothiazole-based dual VEGFR-2/EGFR inhibitors targeting breast and liver cancers: Synthesis, cytotoxic activity, QSAR and molecular docking studies.Bioorg Med Chem Lett. 2022 Feb 15;58:128529. doi: 10.1016/j.bmcl.2022.128529. Epub 2022 Jan 7. Bioorg Med Chem Lett. 2022. PMID: 35007724 Review.
-
Identification of Benzothiazoles Bearing 1,3,4-Thiadiazole as Antiproliferative Hybrids Targeting VEGFR-2 and BRAF Kinase: Design, Synthesis, BIO Evaluation and In Silico Study.Molecules. 2024 Jul 4;29(13):3186. doi: 10.3390/molecules29133186. Molecules. 2024. PMID: 38999138 Free PMC article.
-
Design, synthesis, molecular docking, anticancer evaluations, and in silico pharmacokinetic studies of novel 5-[(4-chloro/2,4-dichloro)benzylidene]thiazolidine-2,4-dione derivatives as VEGFR-2 inhibitors.Arch Pharm (Weinheim). 2021 Feb;354(2):e2000279. doi: 10.1002/ardp.202000279. Epub 2020 Oct 19. Arch Pharm (Weinheim). 2021. PMID: 33073374
-
Targeting the interplay between MMP-2, CA II and VEGFR-2 via new sulfonamide-tethered isomeric triazole hybrids; Microwave-assisted synthesis, computational studies and evaluation.Bioorg Chem. 2022 Jul;124:105816. doi: 10.1016/j.bioorg.2022.105816. Epub 2022 Apr 16. Bioorg Chem. 2022. PMID: 35489270 Review.
Cited by
-
Development of new thiazolidine-2,4-dione hybrids as aldose reductase inhibitors endowed with antihyperglycaemic activity: design, synthesis, biological investigations, and in silico insights.J Enzyme Inhib Med Chem. 2023 Dec;38(1):2231170. doi: 10.1080/14756366.2023.2231170. J Enzyme Inhib Med Chem. 2023. PMID: 37470409 Free PMC article.
-
In-silico Studies, Design, and Synthesis of Pyrimidine-linked Benzothiazoles for its Anticonvulsant Potential.Curr Drug Discov Technol. 2025;22(2):e070524229730. doi: 10.2174/0115701638299019240418055933. Curr Drug Discov Technol. 2025. PMID: 38715333
-
Facile synthesis of hydantoin/1,2,4-oxadiazoline spiro-compounds via 1,3-dipolar cycloaddition of nitrile oxides to 5-iminohydantoins.Beilstein J Org Chem. 2025 Jul 31;21:1552-1560. doi: 10.3762/bjoc.21.118. eCollection 2025. Beilstein J Org Chem. 2025. PMID: 40761763 Free PMC article.
-
Design, synthesis, and in vitro and in vivo biological evaluation of triazolopyrimidine hybrids as multitarget directed anticancer agents.RSC Adv. 2024 Nov 7;14(48):35239-35254. doi: 10.1039/d4ra06704e. eCollection 2024 Nov 4. RSC Adv. 2024. PMID: 39512645 Free PMC article.
-
Synthesis, cytotoxicity and 99mTc-MIBI tumor cell uptake evaluation of 2-phenylbenzothiazole tagged triazole derivatives.Future Med Chem. 2024;16(19):1999-2012. doi: 10.1080/17568919.2024.2389771. Epub 2024 Sep 4. Future Med Chem. 2024. PMID: 39229781
References
-
- Varmus H. The new era in cancer research. Science. 2006;312(5777):1162–1165. - PubMed
-
- Wang Z, Shi X-H, Wang J, Zhou T, Xu Y-Z, Huang T-T, Li Y-F, Zhao Y-L, Yang L, Yang S-Y.. Synthesis, structure–activity relationships and preliminary antitumor evaluation of benzothiazole-2-thiol derivatives as novel apoptosis inducers. Bioorganic Med Chem Lett. 2011;21:1097–1101. - PubMed
-
- Hamdi A, Said E, Farahat AA, Aa El-Bialy S, Am Massoud M.. Synthesis and in vivo antifibrotic activity of novel leflunomide analogues. Lett Drug Des Discov. 2016;13:912–920.
-
- Evan GI, Vousden KH.. Proliferation, cell cycle and apoptosis in cancer. Nature. 2001;411(6835):342–348. - PubMed
-
- Branca MA. Multi-kinase inhibitors create buzz at ASCO. Nat Biotechnol. 2005;23(6):639–640. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Other Literature Sources
