Protocol for a MULTI-centre feasibility study to assess the use of 99mTc-sestaMIBI SPECT/CT in the diagnosis of kidney tumours (MULTI-MIBI study)
- PMID: 36693694
- PMCID: PMC9884914
- DOI: 10.1136/bmjopen-2022-067496
Protocol for a MULTI-centre feasibility study to assess the use of 99mTc-sestaMIBI SPECT/CT in the diagnosis of kidney tumours (MULTI-MIBI study)
Abstract
Introduction: The incidence of renal tumours is increasing and anatomic imaging cannot reliably distinguish benign tumours from renal cell carcinoma. Up to 30% of renal tumours are benign, with oncocytomas the most common type. Biopsy has not been routinely adopted in many centres due to concerns surrounding non-diagnostic rate, bleeding and tumour seeding. As a result, benign masses are often unnecessarily surgically resected. 99mTc-sestamibi SPECT/CT has shown high diagnostic accuracy for benign renal oncocytomas and other oncocytic renal neoplasms of low malignant potential in single-centre studies. The primary aim of MULTI-MIBI is to assess feasibility of a multicentre study of 99mTc-sestamibi SPECT/CT against a reference standard of histopathology from surgical resection or biopsy. Secondary aims of the study include obtaining estimates of 99mTc-sestamibi SPECT/CT sensitivity and specificity and to inform the design and conduct of a future definitive trial.
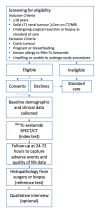
Methods and analysis: A feasibility prospective multicentre study of participants with indeterminate, clinical T1 renal tumours to undergo 99mTc-sestamibi SPECT/CT (index test) compared with histopathology from biopsy or surgical resection (reference test). Interpretation of the index and reference tests will be blinded to the results of the other. Recruitment rate as well as estimates of sensitivity, specificity, positive and negative predictive value will be reported. Semistructured interviews with patients and clinicians will provide qualitative data to inform onward trial design and delivery. Training materials for 99mTc-sestamibi SPECT/CT interpretation will be developed, assessed and optimised. Early health economic modelling using a decision analytic approach for different diagnostic strategies will be performed to understand the potential cost-effectiveness of 99mTc-sestamibi SPECT/CT.
Ethics and dissemination: Ethical approval has been granted (UK HRA REC 20/YH/0279) protocol V.5.0 dated 21/6/2022. Study outputs will be presented and published nationally and internationally.
Trial registration number: ISRCTN12572202.
Keywords: HEALTH ECONOMICS; Nuclear radiology; Urological tumours.
© Author(s) (or their employer(s)) 2023. Re-use permitted under CC BY. Published by BMJ.
Conflict of interest statement
Competing interests: GDS has received educational grants from Pfizer, AstraZeneca and Intuitive Surgical; consultancy fees from Pfizer, Merck, EUSA Pharma and CMR Surgical; Travel expenses from Pfizer and Speaker fees from Pfizer. SD provides educational consultancy for GE Healthcare, Bayer, AAA and AVAANT diagnostics.
Similar articles
-
Cost-effectiveness Analysis of 99mTc-sestamibi SPECT/CT to Guide Management of Small Renal Masses.Eur Urol Focus. 2021 Jul;7(4):827-834. doi: 10.1016/j.euf.2020.02.010. Epub 2020 Feb 27. Eur Urol Focus. 2021. PMID: 32115400
-
Prospective Evaluation of (99m)Tc-sestamibi SPECT/CT for the Diagnosis of Renal Oncocytomas and Hybrid Oncocytic/Chromophobe Tumors.Eur Urol. 2016 Mar;69(3):413-6. doi: 10.1016/j.eururo.2015.08.056. Epub 2015 Sep 18. Eur Urol. 2016. PMID: 26386607
-
Diagnostic Accuracy of 99mTc-Sestamibi SPECT/CT for Characterization of Solid Renal Masses.J Nucl Med. 2023 Jan;64(1):90-95. doi: 10.2967/jnumed.122.264329. Epub 2022 Jun 30. J Nucl Med. 2023. PMID: 35772963
-
99mTc-sestamibi SPECT/CT for the characterization of renal masses: a pictorial guide.Br J Radiol. 2018 Apr;91(1084):20170526. doi: 10.1259/bjr.20170526. Epub 2018 Feb 27. Br J Radiol. 2018. PMID: 29271233 Free PMC article. Review.
-
Characterizing Indeterminate Renal Masses with Molecular Imaging: the Role of 99mTc-MIBI SPECT/CT.Curr Urol Rep. 2017 Sep 12;18(11):86. doi: 10.1007/s11934-017-0737-0. Curr Urol Rep. 2017. PMID: 28900880 Review.
Cited by
-
Diagnostic Biopsy for Small Renal Tumours: A Survey of Current European Practice.Eur Urol Open Sci. 2024 Mar 2;62:54-60. doi: 10.1016/j.euros.2024.02.002. eCollection 2024 Apr. Eur Urol Open Sci. 2024. PMID: 38585205 Free PMC article.
-
99mTc-sestamibi SPECT/CT-the jury is still out!Transl Androl Urol. 2024 Jun 30;13(6):1053-1055. doi: 10.21037/tau-23-623. Epub 2024 May 13. Transl Androl Urol. 2024. PMID: 38983466 Free PMC article. No abstract available.
-
Machine Learning Integrating 99mTc Sestamibi SPECT/CT and Radiomics Data Achieves Optimal Characterization of Renal Oncocytic Tumors.Cancers (Basel). 2023 Jul 9;15(14):3553. doi: 10.3390/cancers15143553. Cancers (Basel). 2023. PMID: 37509214 Free PMC article.
-
Imaging modalities for characterising T1 renal tumours: A systematic review and meta-analysis of diagnostic accuracy.BJUI Compass. 2024 Jun 21;5(7):636-650. doi: 10.1002/bco2.355. eCollection 2024 Jul. BJUI Compass. 2024. PMID: 39022655 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials