Intravenous immunoglobulin for the treatment of Kawasaki disease
- PMID: 36695415
- PMCID: PMC9875364
- DOI: 10.1002/14651858.CD014884.pub2
Intravenous immunoglobulin for the treatment of Kawasaki disease
Abstract
Background: Kawasaki disease (KD) is an acute systemic vasculitis (inflammation of the blood vessels) that mainly affects children. Symptoms include fever, chapped lips, strawberry tongue, red eyes (bulbar conjunctival injection), rash, redness, swollen hands and feet or skin peeling; and enlarged cervical lymph nodes. High fevers and systemic inflammation characterise the acute phase. Inflammation of the coronary arteries causes the most serious complication of the disease, coronary artery abnormalities (CAAs). The primary treatment is intravenous immunoglobulin (IVIG) and acetylsalicylic acid (ASA/aspirin), with doses and regimens differing between institutions. It is important to know which regimens are the safest and most effective in preventing complications.
Objectives: To evaluate the efficacy and safety of IVIG in treating and preventing cardiac consequences of Kawasaki disease.
Search methods: The Cochrane Vascular Information Specialist searched the Cochrane Vascular Specialised Register, CENTRAL, MEDLINE, Embase, and CINAHL databases, and the World Health Organization International Clinical Trials Registry Platform and ClinicalTrials.gov trials registers to 26 April 2022.
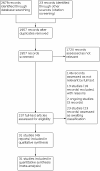
Selection criteria: We included randomised controlled trials (RCTs) investigating the use of IVIG for the treatment of KD. We included studies involving treatment for initial or refractory KD, or both.
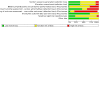
Data collection and analysis: We used standard Cochrane methods. Our primary outcomes were incidence of CAAs and incidence of any adverse effects after treatment. Our secondary outcomes were acute coronary syndromes, duration of fever, need for additional treatment, length of hospital stay, and mortality. We used GRADE to assess the certainty of the evidence for each outcome.
Main results: We identified 31 RCTs involving a total of 4609 participants with KD. Studies compared IVIG with ASA, another dose or regimen of IVIG, prednisolone, or infliximab. The majority of studies reported on primary treatment, so those results are reported below. A limited number of studies investigated secondary or tertiary treatment in IVIG-resistant patients. Doses and regimens of IVIG infusion varied between studies, and all studies had some concerns related to risk of bias. Primary treatment with IVIG compared to ASA for people with KD Compared to ASA treatment, IVIG probably reduces the incidence of CAAs in people with KD up to 30 days (odds ratio (OR) 0.60, 95% confidence interval (CI) 0.41 to 0.87; 11 studies, 1437 participants; moderate-certainty evidence). The individual studies reported a range of adverse effects, but there was little to no difference in numbers of adverse effects between treatment groups (OR 0.57, 95% CI 0.17 to 1.89; 10 studies, 1376 participants; very low-certainty evidence). There was limited evidence for the incidence of acute coronary syndromes, so we are uncertain of any effects. Duration of fever days from treatment onset was probably shorter in the IVIG group (mean difference (MD) -4.00 days, 95% CI -5.06 to -2.93; 3 studies, 307 participants; moderate-certainty evidence). There was little or no difference between groups in need for additional treatment (OR 0.27, 95% CI 0.05 to 1.57; 3 studies, 272 participants; low-certainty evidence). No study reported length of hospital stay, and no deaths were reported in either group. Primary treatment with IVIG compared to different infusion regimens of IVIG for people with KD Higher-dose regimens of IVIG probably reduce the incidence of CAAs compared to medium- or lower-dose regimens of IVIG up to 30 days (OR 0.60, 95% CI 0.40 to 0.89; 8 studies, 1824 participants; moderate-certainty evidence). There was little to no difference in the number of adverse effects between groups (OR 1.11, 95% CI 0.52 to 2.37; 6 studies, 1659 participants; low-certainty evidence). No study reported on acute coronary syndromes. Higher-dose IVIG may reduce the duration of fever compared to medium- or lower-dose regimens (MD -0.71 days, 95% CI -1.36 to -0.06; 4 studies, 992 participants; low-certainty evidence). Higher-dose regimens may reduce the need for additional treatment (OR 0.29, 95% CI 0.10 to 0.88; 4 studies, 1125 participants; low-certainty evidence). We did not detect a clear difference in length of hospital stay between infusion regimens (MD -0.24, 95% CI -0.78 to 0.30; 3 studies, 752 participants; low-certainty evidence). One study reported mortality, and there was little to no difference detected between regimens (moderate-certainty evidence). Primary treatment with IVIG compared to prednisolone for people with KD The evidence comparing IVIG with prednisolone on incidence of CAA is very uncertain (OR 0.60, 95% CI 0.24 to 1.48; 2 studies, 140 participants; very low-certainty evidence), and there was little to no difference between groups in adverse effects (OR 4.18, 95% CI 0.19 to 89.48; 1 study; 90 participants; low-certainty evidence). We are very uncertain of the impact on duration of fever, as two studies reported this outcome differently and showed conflicting results. One study reported on acute coronary syndromes and mortality, finding little or no difference between groups (low-certainty evidence). No study reported the need for additional treatment or length of hospital stay.
Authors' conclusions: The included RCTs investigated a variety of comparisons, and the small number of events observed during the study periods limited detection of effects. The certainty of the evidence ranged from moderate to very low due to concerns related to risk of bias, imprecision, and inconsistency. The available evidence indicated that high-dose IVIG regimens are probably associated with a reduced risk of CAA formation compared to ASA or medium- or low-dose IVIG regimens. There were no clinically significant differences in incidence of adverse effects, which suggests there is little concern about the safety of IVIG. Compared to ASA, high-dose IVIG probably reduced the duration of fever, but there was little or no difference detected in the need for additional treatment. Compared to medium- or low-dose IVIG, there may be reduced duration of fever and reduced need for additional treatment. We were unable to draw any conclusions regarding acute coronary syndromes, mortality, or length of hospital stay, or for the comparison IVIG versus prednisolone. Our findings are in keeping with current guideline recommendations and evidence from long-term epidemiology studies.
Copyright © 2023 The Authors. Cochrane Database of Systematic Reviews published by John Wiley & Sons, Ltd. on behalf of The Cochrane Collaboration.
Conflict of interest statement
CB: none known. As CB is based within Cochrane Vascular, editorial tasks for this review update were carried out by other members of the Cochrane Vascular editorial team. SK: none known MS: none known SI: none known TK: declares that his institution received a scholarship grant from Japan Blood Products Organization (total amount JPY 300,000 in 2021). This does not conflict with his work on this review. TK also reports payments for lectures from Japan Blood Products Organization, Mitsubishi Tanabe Pharma Corporation, Nihon Pharmaceutical, and Teijin Pharma.
Figures






































Update of
- doi: 10.1002/14651858.CD014884
Similar articles
-
Systemic interventions for treatment of Stevens-Johnson syndrome (SJS), toxic epidermal necrolysis (TEN), and SJS/TEN overlap syndrome.Cochrane Database Syst Rev. 2022 Mar 11;3(3):CD013130. doi: 10.1002/14651858.CD013130.pub2. Cochrane Database Syst Rev. 2022. PMID: 35274741 Free PMC article.
-
Drugs for preventing postoperative nausea and vomiting in adults after general anaesthesia: a network meta-analysis.Cochrane Database Syst Rev. 2020 Oct 19;10(10):CD012859. doi: 10.1002/14651858.CD012859.pub2. Cochrane Database Syst Rev. 2020. PMID: 33075160 Free PMC article.
-
Preoperative coronary interventions for preventing acute myocardial infarction in the perioperative period of major open vascular or endovascular surgery.Cochrane Database Syst Rev. 2024 Jul 3;7(7):CD014920. doi: 10.1002/14651858.CD014920.pub2. Cochrane Database Syst Rev. 2024. PMID: 38958136 Free PMC article.
-
Non-pharmacological interventions for preventing delirium in hospitalised non-ICU patients.Cochrane Database Syst Rev. 2021 Jul 19;7(7):CD013307. doi: 10.1002/14651858.CD013307.pub2. Cochrane Database Syst Rev. 2021. PMID: 34280303 Free PMC article.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2017 Dec 22;12(12):CD011535. doi: 10.1002/14651858.CD011535.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Jan 9;1:CD011535. doi: 10.1002/14651858.CD011535.pub3. PMID: 29271481 Free PMC article. Updated.
Cited by
-
Machine learning for early diagnosis of Kawasaki disease in acute febrile children: retrospective cross-sectional study in China.Sci Rep. 2025 Feb 25;15(1):6799. doi: 10.1038/s41598-025-90919-y. Sci Rep. 2025. PMID: 40000757 Free PMC article.
-
Basophils Predict Mite Sensitization in Patients with Kawasaki Disease.Children (Basel). 2023 Jul 12;10(7):1209. doi: 10.3390/children10071209. Children (Basel). 2023. PMID: 37508706 Free PMC article.
-
An infant with IVIG-resistant Kawasaki disease requiring repeated glucocorticoids therapy: A case report and literature review.Medicine (Baltimore). 2025 May 23;104(21):e42572. doi: 10.1097/MD.0000000000042572. Medicine (Baltimore). 2025. PMID: 40419879 Free PMC article. Review.
-
Expression of Oxidative Stress and Inflammatory Indicators for Coronary Artery Disease in Kawasaki Disease.Mediterr J Hematol Infect Dis. 2024 Jul 1;16(1):e2024052. doi: 10.4084/MJHID.2024.052. eCollection 2024. Mediterr J Hematol Infect Dis. 2024. PMID: 38984102 Free PMC article.
-
[Evidence-based guidelines for the diagnosis and treatment of Kawasaki disease in children in China (2023)].Zhongguo Dang Dai Er Ke Za Zhi. 2023 Dec 15;25(12):1198-1210. doi: 10.7499/j.issn.1008-8830.2309038. Zhongguo Dang Dai Er Ke Za Zhi. 2023. PMID: 38112136 Free PMC article. Chinese.
References
References to studies included in this review
Barron 1990 {published data only}
-
- Barron KS, Murphy DJ Jr, Silverman ED, Ruttenberg HD, Wright GB, Franklin W, et al. Treatment of Kawaski syndrome: a comparison of two dosage regimens of intravenously administered immune globulin. Journal of Pediatrics 1990;117(4):638-44. - PubMed
Burns 2008 {published data only}
-
- NCT00271570. Infliximab (Remicade) for patients with acute Kawasaki disease. clinicaltrials.gov/show/NCT00271570 (first received 2 January 2006).
Burns 2021 {published data only}
-
- Burns JC, Roberts SC, Tremoulet AH, He F, Printz BF, Ashouri N, et al. Infliximab versus second intravenous immunoglobulin for treatment of resistant Kawasaki disease in the USA (KIDCARE): a randomised, multicentre comparative effectiveness trial. Lancet Child and Adolescent Health 2021;5(12):852-61. - PubMed
-
- NCT03065244. KIDCARE (Kawasaki Disease Comparative Effectiveness Trial). clinicaltrials.gov/show/NCT03065244 (first received 27 February 2017).
-
- Roberts SC, Jain S, Tremoulet AH, Kim KK, Burns JC, KIDCARE Multicenter Study Group. The Kawasaki Disease Comparative Effectiveness (KIDCARE) trial: a phase III, randomized trial of second intravenous immunoglobulin versus infliximab for resistant Kawasaki disease. Contemporary Clinical Trials 2019;79:98-103. - PubMed
Furusho 1984 {published data only}
-
- Furusho K, Kamiya T, Nakano H. High-dose intravenous gammaglobulin for Kawasaki disease. Lancet 1984;2(8411):1055-8. - PubMed
Furusho 1991A {published data only}
Furusho 1991B {published data only}
-
- Furusho K, Kamiya T, Nakano H, Kiyosawa N, Shinomiya K, Hayashidera T, et al. Intravenous gamma-globulin for Kawasaki disease. Acta Paediatrica Japonica: Overseas edition 1991;33(6):799-804. - PubMed
Harada 1989 {published data only}
-
- Harada H, Yamaguchi H. Intravenous gamma globulin treatment in Kawasaki disease - comparison between high dose and low dose. Progress in Medicine 1989;9:45-8.
-
- Harada K. Intravenous gamma-globulin treatment in Kawasaki disease. Acta Paediatrica Japonica: Overseas edition 1991;33(6):805-10. - PubMed
Hashino 2001 {published data only}
-
- Hashino K, Ishii M, Iemura M, Akagi T, Kato H. Re-treatment for immune globulin-resistant Kawasaki disease: a comparative study of additional immune globulin and steroid pulse therapy. Pediatrics International 2001;43(3):211-7. - PubMed
He 2021 {published data only}
-
- He L, Liu F, Huang G. Multi centeral study on IVIG treatment to Kawasaki disease. Cardiology in the Young 2017;27(4):S472.
-
- He L, Niu CW, Liu F, Huang GY, Wu L, Chu C, et al. A prospective randomised study of intravenous immunoglobulin treatment regimens in acute Kawasaki disease. Journal of the Hong Kong College of Cardiology 2016;24(1):49.
-
- NCT02439996. Different doses of IVIG for Kawasaki disease. clinicaltrials.gov/show/NCT02439996 (first received 12 May 2015).
Matsushima 1985 {published data only}
-
- Matsushima M, Nagashima M, Matsuoka H. High dose immunoglobulin therapy for Kawasaki disease by a control study. Nihon Shounika Gakkai Zasshi [Journal of the Japanese Pediatric Society] 1986;90:80-7.
Miura 2008 {published data only}
-
- Miura M, Kohno K, Ohki H, Yoshiba S, Sugaya A, Satoh M. Effects of methylprednisolone pulse on cytokine levels in Kawasaki disease patients unresponsive to intravenous immunoglobulin. European Journal of Pediatrics 2008;167(10):1119-23. - PubMed
Mori 2017 {published data only}
-
- Mori M. Clinical trial of the infliximab for Kawasaki disease that an effect is inadequate for by existing treatment in Japan. Rheumatology (United Kingdom) 2017;56(Suppl 3):P1-67.
-
- NCT01596335. Clinical study of TA-650 in patients with refractory Kawasaki disease. clinicaltrials.gov/ct2/show/NCT01596335 (first received 26 September 2018).
Morikawa 1994 {published data only}
-
- Morikawa Y, Ohashi Y, Harada K, Asai T, Okawa S, Nagashima M, et al. A multicenter, randomized, controlled trial of intravenous gamma globulin therapy in children with acute Kawasaki disease. Acta Paediatrica Japonica: Overseas edition 1994;36(4):347-54. - PubMed
-
- Morikawa Y, Ohashi Y, Harada K, Asai T, Okawa S, Nagashima M, et al. Coronary risks after high-dose gamma-globulin in children with Kawasaki disease. Pediatrics International 2000;42(5):464-9. - PubMed
Nagashima 1987 {published data only}
-
- Nagashima M, Matsushima M, Matsuoka H. High-dose gammaglobulin therapy for Kawasaki disease. Journal of Pediatrics 1987;110(5):710-2. - PubMed
Newburger 1986 {published data only}
-
- Newburger JW, Takahashi M, Burns JC. The treatment of Kawasaki syndrome with intravenous gamma globulin. New England Journal of Medicine 1986;315(6):341-7. - PubMed
-
- Takahashi M, Newburger JW. Long-term follow-up of coronary abnormalities in Kawasaki syndrome treated with and without IV gamma globulin. Circulation 1990;82:717.
Newburger 1991 {published data only}
-
- NCT00000520. Prevention of coronary aneurysms in Kawasaki syndrome. clinicaltrials.gov/show/NCT00000520 (first received 28 October 1999).
-
- Newburger JW, Takahashi M, Beiser AS, Burns JC, Bastian J, Chung KJ, et al. A single intravenous infusion of gamma globulin as compared with four infusions in the treatment of acute Kawasaki syndrome. New England Journal of Medicine 1991;324(23):1633-9. - PubMed
Nishihara 1988 {published data only}
-
- Nishihara S, Ishibashi K, Matsuda I. Intravenous gammaglobulin and reduction of coronary artery abnormalities in children with Kawasaki disease. Lancet 1988;2(8617):973. - PubMed
Nonaka 1995 {published data only}
-
- Nonaka Z, Maekawa K, Okabe T, Eto Y, Kubo M. Randomized controlled study of intravenous prednisolone and gammaglobulin treatment in 100 cases with Kawasaki disease. Kawasaki Disease 1995:328-31.
Ogawa 1987 {published data only}
-
- Ogawa M, Ogino H, Harima Y, Kono S, Ohkuni H, Nishida M. High dose gamma-globulin therapy for Kawasaki disease. Journal of the Japanese Pediatric Society 1987;91(12):3763-71.
-
- Ogino H, Ogawa M, Harima Y, Kono S, Ohkuni H, Nishida M, et al. Clinical evaluation of gammaglobulin preparations for the treatment of Kawasaki disease. Progress in Clinical and Biological Research 1987;250:555-6. - PubMed
-
- Ogino H, Ogawa M, Harima Y, Kono S, Ohkuni H, Nishida M, et al. High-dose intravenous gammaglobulin treatment of Kawasaki disease. Progress in Medicine 1990;10:29-38.
Ogino 1987 {published data only}
-
- Ogino H, Ogawa M, Harima Y, Kono S, Ohkuni H, Nishida M, et al. Clinical evaluation of gammaglobulin preparations for the treatment of Kawasaki disease. Progress in Clinical and Biological Research 1987;250:555-6. - PubMed
-
- Ogino H, Ogawa M, Harima Y, Kono S, Ohkuni H, Nishida M, et al. High-dose intravenous gammaglobulin treatment of Kawasaki disease. Progress in Medicine 1990;10:29-38.
-
- Ogino H, Ogawa M, Harima Y, Kono S, Ohkuni H, Nisida M. Clinical evaluation of gamma globulin preparations for the treatment of Kawasaki disease - trial of 200 mg/kg/day. Journal of the Japanese Pediatric Society 1987;91(8):2849-56.
Okuni 1987A {published data only}
-
- Harada K. Intravenous gamma-globulin treatment in Kawasaki disease. Acta Paediatrica Japonica: Overseas edition 1991;33(6):805-10. - PubMed
-
- Okuni M, Harada K, Yamaguchi H, Yanagawa H, Sonobe T, Kawasaki T. Intravenous gamma globulin therapy in Kawasaki disease - trial of low dose gammaglobulin. Kawasaki Disease 1987:433-9. - PubMed
Okuni 1987B {published data only}
-
- Harada K. Intravenous gamma-globulin treatment in Kawasaki disease. Acta Paediatrica Japonica: Overseas edition 1991;33(6):805-10. - PubMed
-
- Okuni M, Harada K, Yamaguchi H, Yanagawa H, Sonobe T, Kawasaki T. Intravenous gamma globulin therapy in Kawasaki disease - trial of low dose gammaglobulin. Kawasaki Disease 1987:433-9. - PubMed
Onouchi 1988 {published data only}
-
- Onouchi Z, Isogai Y, Yanagisawa M, Yamanouchi T, Matsuda H, Morita T, et al. Multicenter randomized controlled study of intravenous immunoglobulin in Kawasaki disease. Journal of the Japanese Pediatric Society 1988;92(11):2367-76.
-
- Onouchi Z, Yanagisawa M, Hirayama T, Kiyosawa N, Matsuda H, Nakashima M. Optimal dosage and differences in therapeutic efficacy of IGIV in Kawasaki disease. Acta Paediatrica Japonica: Overseas edition 1995;37(1):40-6. - PubMed
Onouchi 1992 {published data only}
-
- Onouchi Z, Yanagisawa M, Hirayama T, Kiyosawa N, Matsuda H, Nakashima M. Optimal dosage and differences in therapeutic efficacy of IGIV in Kawasaki disease. Acta Paediatrica Japonica: Overseas edition 1995;37(1):40-6. - PubMed
-
- Onouchi Z, Yanagisawa M, Shiraishi H, Hirayama T, Katsube Y, Kiyosawa N, et al. Multi-centre randomized controlled study of intravenous immunoglobulin G (C-425) for Kawasaki disease. Journal of the Japanese Pediatric Society 1992;96(12):2669-79.
Qin 2006 {published data only}
-
- Qin LJ, Wang HW, Hu XF, Liu QJ, Shi H, Wei YX, et al. Therapeutic effectiveness of intravenous immunoglobulin at 1 g/kg and 2 g/kg on Kawasaki disease: a comparative and follow-up study. Zhonghua er ke za zhi 2006;44(12):891-5. - PubMed
Sakata 2007 {published data only}
-
- Ozawa S, Sakata K, Hamaoka K. Randomized-prospective study of a single infusion of 1g/kg gammaglobulin for reducing a total dose of gammaglobulin in Kawasaki disease treatment. Pediatric Research 2003;53:181.
-
- Sakata K, Hamaoka K, Ozawa SI, Niboshi A, Yoshihara T, Nishiki T, et al. A randomized prospective study on the use of 2 g-IVIG or 1 g-IVIG as therapy for Kawasaki disease. European Journal of Pediatrics 2007;166(6):565-71. - PubMed
Sato 1995 {published data only}
-
- Sato N, Sugimura T, Akagi T, Inoue O, Ono E, Kato H. Selective gammaglobulin treatment in Kawasaki disease: single 2 g/kg or 400 mg/kg/day for 5 days? Kawasaki Disease 1995:332-8.
-
- Sato N, Sugimura T, Akagi T, Yamakawa R, Hashino K, Eto G, et al. Selective high dose gamma-globulin treatment in Kawasaki disease: assessment of clinical aspects and cost effectiveness. Pediatrics International 1999;41(1):1-7. - PubMed
Wang 2020 {published data only}
-
- ChiCTR-EOC-17013266. The mechanism of coronary artery lesions and effect of treatment in children with Kawasaki disease: a clinical cohort study. chictr.org.cn/showproj.aspx?proj=22637 (first received 11 June 2017).
Yabiku 1989 {published data only}
-
- Ogino H, Ogawa M, Harima Y, Kono S, Ohkuni H, Nishida M, et al. High-dose intravenous gammaglobulin treatment of Kawasaki disease. Progress in Medicine 1990;10:29-38.
-
- Yabiku M, Kojima S, Ogawa M, Ogino H, Harima Y, Kono S, et al. High dose gammaglobulin therapy for Kawasaki disease. Journal of the Japanese Pediatric Society 1989;93(12):2721-31.
Youn 2016 {published data only}
-
- Youn Y, Kim J, Hong YM, Sohn S. Infliximab as the first retreatment in patients with Kawasaki disease resistant to initial intravenous immunoglobulin. Pediatric Infectious Disease Journal 2016;35(4):457-9. - PubMed
Yuan 2000 {published data only}
-
- Yuan Y. Analysis of recent effects for corticosteroid in treating Kawasaki disease. Chinese Journal of Practical Paediatrics 2000;15:49.
References to studies excluded from this review
ChiCTR2000035163 {published data only}
-
- ChiCTR2000035163. A multicenter clinical trial of a new individualized treatment opinion for Kawasaki disease. chictr.org.cn/showprojen.aspx?proj=57061 (first received 2 August 2020).
Hamada 2019 {published data only}
-
- Hamada H, Suzuki H, Onouchi Y, Ebata R, Terai M, Fuse S, et al. Efficacy of primary treatment with immunoglobulin plus ciclosporin for prevention of coronary artery abnormalities in patients with Kawasaki disease predicted to be at increased risk of non-response to intravenous immunoglobulin (KAICA): a randomised controlled, open-label, blinded-endpoints, phase 3 trial. Lancet 2019;393(10176):1128-37. - PubMed
-
- Hamada H, Suzuki H, Onouchi Y, Ebata R, Terai M, Fuse S, et al. Efficacy of primary treatment with immunoglobulin plus cyclosporine for prevention of coronary artery abnormalities in Kawasaki disease patients predicted to be at increased risk of IVIG non-response (KAICA study): a controlled, phase 3, randomised, open-label, blinded-endpoints trial. Rheumatology 2019;58(Suppl 2):kez063-031. [DOI: 10.1093/rheumatology/kez063.031] - DOI - PubMed
ISRCTN71987471 {published data only}
-
- ISRCTN71987471. Corticosteroids plus standard of care treatment versus standard of care treatment alone to prevent heart complications in Kawasaki disease. isrctn.com/ISRCTN71987471 (first received 31 March 2020).
JPRN‐UMIN000014665 {published data only}
-
- JPRN-UMIN000014665. The investigation of the therapeutic efficacy of rapid intravenous immunoglobulin injection for acute phase Kawasaki disease. trialsearch.who.int/Trial2.aspx?TrialID=JPRN-UMIN000014665 (first received 30 October 2014).
Lee 1996 {published data only}
-
- Lee HK, Kim DS, Noh GW, Lee KY. Effects of intravenous immune globulin on the peripheral lymphocyte phenotypes in Kawasaki disease. Yonsei Medical Journal 1996;37(5):357-63. - PubMed
Lin 2021 {published data only}
-
- Lin SY, He L, Xie LP, Wang Y, Lin YX, Cao YY, et al. Effects of immunoglobulin plus prednisolone in reducing coronary artery lesions in patients with Kawasaki disease: study protocol for a phase III multicenter, open-label, blinded-endpoints randomized controlled trial. Trials 2021;22(2):898. - PMC - PubMed
Muta 2002 {published data only}
-
- Muta H, Ishii M, Hirose A, Furui J, Sugahara Y, Himeno W, et al. Randomized prospective controlled study of intravenous gamma-globulin products in treatment of Kawasaki disease. Journal of the Japan Pediatric Society 2002;106(6):742-6.
-
- Muta H, Ishii M, Sugahara Y, Akagi T, Kato H. Randomized controlled study of intravenous gamma-globulin products in treatment of Kawasaki disease. Pediatric Research 2003;53(1):181.
Nanishi 2017 {published data only}
-
- Nanishi E, Nishio H, Takada H, Yamamura K, Fukazawa M, Furuno K, et al. Clarithromycin plus intravenous immunoglobulin therapy can reduce the relapse rate of Kawasaki disease: a phase 2, open-label, randomized control study. Journal of the American Heart Association 2017;6(7):e005370. - PMC - PubMed
NCT02298062 {published data only}
-
- NCT02298062. Infliximab for Kawasaki disease patients resistant to IVIG: a multicentre, prospective, randomised trial. clinicaltrials.gov/ct2/show/NCT02298062 (first received 21 November 2014).
Portman 2019 {published data only}
-
- Portman MA, Dahdah N, Olson AK, Slee A, Choueiter N, Altman C. Multicenter, randomized, double-blind trial of etanercept in acute Kawasaki disease. Circulation 2017;136:A15447.
RAISE 2012 {published data only}
-
- Kobayashi T, Saji T, Otani T, Takeuchi K, Nakamura T, Arakawa H, et al. Efficacy of immunoglobulin plus prednisolone for prevention of coronary artery abnormalities in severe Kawasaki disease (RAISE study): a randomised, open-label, blinded-endpoints trial. Lancet 2012;379(9826):1613-20. [DOI: 10.1016/S0140-6736(11)61930-2] - DOI - PubMed
Sanati 2021 {published data only}
Seki 2021 {published data only}
References to studies awaiting assessment
Chang 2007 {published data only}
-
- Chang YC. Two different large dosage of gamma-immunoglobulin for the treatment of Kawasaki disease. Sichuan Medical Journal 2007;28(7):801.
Chang 2008 {published data only}
-
- Chang WH. The different dosage of gamma-immunoglobulin in treating of Kawasaki disease - a clinical study. Journal of Jilin Medical College 2008;29(4):199-201.
Chen 2006 {published data only}
-
- Chen PC, Shi WZ. Medium dose of gamma-immunoglobulin for the treatment of Kawasaki disease - a clinical observation. Zhejiang Clinical Medicine 2006;8(3):308.
Fu 2010 {published data only}
-
- Fu CN, Chen HP. Comparison of the therapeutic effects of different dose intravenous gamma-immunoglobulin in Kawasaki disease. China Modern Drug Application 2010;4(7):121-2.
Ho 2003 {published data only}
-
- Ho HH, Chang Q, Lee JY. Different dosage of intravenous immunoglobulin for the treatment of Kawasaki disease - the observation of therapeutic effect. FJ Medical Journal 2003;25(2):49-50.
Ho 2004 {published data only}
-
- Ho C. Comparison of therapeutic efficacy of two intravenous gamma-immunoglobulin regimen in acute Kawasaki disease. Hebei Medicine 2004;26(4):323.
Juan 2003 {published data only}
-
- Juan HL, Xiang RL, Liang GA, Lu HP. The effect of different IVIG dosage on CD4+ /CD8+ of children with Kawasaki disease. Pediatric Pharmacological Magazine 2003;9(3):18-9.
Juan 2005 {published data only}
-
- Juan C, Yang HB, Juan J, Ho XL. Comparison of therapeutic efficacy of two large dosage intravenous gamma-immunoglobulin regimens in pediatric Kawasaki disease. Practical Clinical Medicine 2005;8(3):48-9.
Juan 2006 {published data only}
-
- Juan C, Cai XJ. Comparison of therapeutic efficacy of two gamma-immunoglobulin regimens in acute Kawasaki disease. Qingdao Medicine and Health 2006;38(3):175-6.
Kuo 2009 {published data only}
-
- Kuo L, Chang HY, Dou JL, Xu FX, Zhai YL. Different dosage of intravenous gamma-immunoglobulin for the treatment of Kawasaki disease. The observation of therapeutic effect and nursing experience. China Practical Clinical Medicine 2009;3(7):116-7.
Li 2008 {published data only}
-
- Li HL, Liao Y. Analysis of the therapeutic efficacy of a single dose gamma-immunoglobulin for the treatment of Kawasaki disease in thirty three children. Journal of Gannan Medical University 2008;28(1):36-8.
Li 2009 {published data only}
-
- Li CK. Two different intravenous gamma-immunoglobulin regimens in the treatment acute Kawasaki disease - an observation of therapeutic efficacy. Practical Clinical Medicine 2009;10(12):60-2.
Liao 2007 {published data only}
-
- Liao J, He KJ. The clinical study of different dosage immune globulin in treating Kawasaki in intravenous injection. China Healthcare Innovation 2007;2(22):8-9.
Liu 2000 {published data only}
-
- Liu HM. The exploration of intravenous immunoglobulin dosage for the treatment of Kawasaki disease. Shantung Medicine 2000;40(8):23-4.
Liu 2004 {published data only}
-
- Liu TC. The observation of therapeutic effect of intravenous gamma-immunoglobulin in Kawasaki disease. Modern Journal of Integrated Traditional Chinese and Western Medicine 2004;13(4):451.
Liu 2009 {published data only}
-
- Liu M, Cheng J, Luo Q. Comparison of therapeutic efficacy of two intravenous gamma-immunoglobulin regimens in children with Kawasaki disease. China Eugenics and Hereditary Magazine 2009;17(2):117-8.
Lu 2003 {published data only}
-
- Lu JX, Zheng ZL. Therapeutic effects of intravenous immune globulin on Kawasaki disease. Oinghai Medical Magazine 2003;33(5):16-8.
Peng 2001 {published data only}
-
- Peng CM, Xiao Q, Chen YL, Su Y. A study on different dose of intravenous immunoglobulins for treating Kawasaki disease. Journal of Nanhua University (Medical Edition) 2001;29(6):581-3.
Teng 2005 {published data only}
-
- Teng MY. Comparison of therapeutic efficacy of two gamma-immunoglobulin regimens in Kawasaki disease. International Medical Health Reports 2005;11(2):33-4.
Yao 2009 {published data only}
-
- Yao SH. The observation of therapeutic efficacy of different intravenous gamma-immunoglobulin for the treatment in sixty-four children with Kawasaki disease. China Community Physician 2009;11(213):68-9.
Yen 2007 {published data only}
-
- Yen P. The observation of therapeutic efficacy of different intravenous gamma-immunoglobulin for the treatment in Kawasaki disease. Community Medical Magazine 2007;5(23):11.
Yuan 2009 {published data only}
-
- Yuan YF, Kuai WX. The application of gamma-immunoglobulin for pediatric Kawasaki disease. Shanxi Medical Magazine 2009;38(4):365-6.
Yueh 2006 {published data only}
-
- Yueh AH, Tian QL, Li H. The observation of therapeutic efficacy of intravenous gamma-immunoglobulin for the treatment in fifty-six children with Kawasaki disease. Clinical Gathering 2006;21(13):962-3.
References to ongoing studies
ChiCTR1900027954 {published data only}
-
- ChiCTR1900027954. Effecacy of infliximab on IVIG-unresponsive Kawasaki disease: a single center prospective randamized controlled study. trialsearch.who.int/Trial2.aspx?TrialID=ChiCTR1900027954 (first received 7 December 2019).
EUCTR2020‐003194‐22‐FR {published data only}
-
- EUCTR2020-003194-22-FR. A randomized phase III multicentre trial comparing the efficacy and safety of Anakinra versus intravenous immunoglobulin (IVIG) retreatment, in patients with Kawasaki disease who failed to respond to initial standard IVIG treatment (ANACOMP) [Essai multicentrique randomisé de phase III comparant l'efficacité et la sécurité de l'anakinra au retraitement par immunoglobulines intraveineuses (IVIG), chez des patients atteints de la maladie de Kawasaki qui n'ont pas répondu au traitement initial standard par IVIG]. trialsearch.who.int/Trial2.aspx?TrialID=EUCTR2020-003194-22-FR (first received 7 September 2020).
-
- NCT04656184. A randomized phase III multicenter trial comparing the efficacy and safety of anakinra versus intravenous immunoglobulin (IVIG) retreatment, in patients with Kawasaki disease who failed to respond to initial standard IVIG treatment. clinicaltrials.gov/show/NCT04656184 (first received 7 December 2020).
Additional references
Ae 2020
Ae 2021
-
- Ae R, Yashiro M, Matsubara Y, Osami M, Makino S, Nakamura Y. Results of the 26th nationwide survey of Kawasaki disease in Japan. jichi.ac.jp/dph/wp-dph/wp-content/uploads/2022/04/a19b047d4b9e6fbb84b6b1....
Ashouri 2008
-
- Ashouri N, Takahashi M, Dorey F, Mason W. Risk factors for non response to therapy in Kawasaki disease. Journal of Pediatrics 2008;153(3):3. - PubMed
Atkins 2004
Ayusawa 2005
-
- Ayusawa M, Sonobe T, Uemura S, Ogawa S, Nakamura Y, Kiyosawa N, Kawasaki Disease Research Committee. Revision of diagnostic guidelines for Kawasaki disease (the 5th revised edition). Pediatrics International 2005;47(2):232-4. - PubMed
Baumer 2006
Burns 1996
-
- Burns JC, Shike H, Gordon JB, Malhotra A, Schoenwetter M, Kawasaki T. Sequelae of Kawasaki disease in adolescents and young adults. Journal of the American College of Cardiology 1996;28:253–7. - PubMed
Burns 2015
ChiCTR‐EOC‐17013266
-
- ChiCTR-EOC-17013266. The mechanism of coronary artery lesions and effect of treatment in children with Kawasaki disease: a clinical cohort study. chictr.org.cn/showproj.aspx?proj=22637 (first received 11 June 2017).
Covidence [Computer program]
-
- Covidence. Version accessed 15 June 2021. Melbourne, Australia: Veritas Health Innovation. Available at covidence.org.
Dallaire 2010
Daniels 2012
Deeks 2021
-
- Deeks JJ, Higgins JP, Altman DG, editor(s). Chapter 10: Analysing data and undertaking meta-analyses. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (updated February 2021). Cochrane, 2021. Available from training.cochrane.org/handbook/archive/v6.2.
de Graeff 2019
-
- Graeff N, Groot N, Ozen S, Eleftheriou D, Avcin T, Bader-Meunier B, et al. European consensus-based recommendations for the diagnosis and treatment of Kawasaki disease - the SHARE initiative. Rheumatology (OXFORD) 2019;58(4):672-82. - PubMed
Franco 2010
Gedalia 2007
GRADEpro GDT [Computer program]
-
- GRADEpro GDT. Version accessed 6 November 2020. Hamilton (ON): McMaster University (developed by Evidence Prime). Available at gradepro.org.
Green 2022
Hamada 2019
-
- Hamada H, Suzuki H, Onouchi Y, Ebata R, Terai M, Fuse S, et al. Efficacy of primary treatment with immunoglobulin plus ciclosporin for prevention of coronary artery abnormalities in patients with Kawasaki disease predicted to be at increased risk of non-response to intravenous immunoglobulin (KAICA): a randomised controlled, open-label, blinded-endpoints, phase 3 trial. Lancet 2019;393(10176):1128–37. - PubMed
Harada 1991
-
- Harada K. Intravenous gamma-globulin treatment in Kawasaki disease. Acta Paediatrica Japonica: Overseas edition 1991;33(6):805-10. - PubMed
Hayasaka 2003
Higgins 2011
-
- Higgins JP, Green S, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 5.1.0 (updated March 2011). The Cochrane Collaboration, 2011. Available from training.cochrane.org/handbook/archive/v5.1/.
Higgins 2021
-
- Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (updated February 2021). Cochrane, 2021. Available from training.cochrane.org/handbook/archive/v6.2.
Hokosaki 2012
Holman 2010
JMHW 1984
-
- Research Committee on Kawasaki Disease. Report of Subcommittee on Standardization of Diagnostic Criteria and Reporting of Coronary Artery Lesions in Kawasaki Disease. Tokyo: Japanese Ministry of Health and Welfare; 1984.
Kawasaki 1967
-
- Kawasaki T. Acute febrile mucocutaneous syndrome with lymphoid involvement with specific desquamation of the fingers and toes in children. Arerugi 1967;16(3):178–222. - PubMed
Kobayashi 2016
-
- Kobayashi T, Fuse S, Sakamoto N, Mikami M, Ogawa S, Hamaoka K, Z Score Project Investigators. A new Z score curve of the coronary arterial internal diameter using the lambda-mu-sigma method in a pediatric population. Journal of the American Society of Echocardiography 2016;29(8):794-801.e29. [DOI: 10.1016/j.echo.2016.03.017] [PMID: ] - DOI - PubMed
Kobayashi 2020
Kone‐Paut 2018
Lee 2008
Lefebvre 2022
-
- Lefebvre C, Glanville J, Briscoe S, Featherstone R, Littlewood A, Marshall C, et al. Chapter 4: Searching for and selecting studies. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editor(s). Cochrane Handbook for Systematic Reviews of Interventions version 6.3 (updated February 2022). Cochrane, 2022. Available from training.cochrane.org/handbook.
Liberati 2009
Maddox 2015
-
- Maddox RA, Person MK, Lindsay JL, Baherling DL, Steiner CA, Schonberger LB, et al. Monitoring the occurrence of Kawasaki syndrome in the United States. Circulation 2015;131(Suppl 2):Abstract 0.03.
Makino 2015
Makino 2019
McCrindle 2017
-
- McCrindle BW, Rowley AH, Newburger JW, Burns JC, Bolger AF, Gewitz M, American Heart Association Rheumatic Fever, Endocarditis, and Kawasaki Disease Committee of the Council on Cardiovascular Disease in the Young, Council on Cardiovascular and Stroke Nursing, Council on Cardiovascular Surgery and Anesthesia, and Council on Epidemiology and Prevention, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a scientific statement for health professionals from the American Heart Association. Circulation 2017;135(17):e927-99. [DOI: 10.1161/CIR.0000000000000484] - DOI - PubMed
Miura 2021
-
- Miura M, Ayusawa M, Fukazawa R, Hamada H, Ikeda S, Ito S, et al. Guidelines for medical treatment of acute Kawasaki disease (2020 revised version). Journal of Pediatric Cardiology and Cardiac Surgery 2021;5(1):41-73. [DOI: 10.24509/jpccs.0501G1] - DOI
Mori 2004
Newburger 1986
-
- Newburger JW, Takahashi M, Burns JC, Beiser AS, Chung KJ, Duffy CE, et al. The treatment of Kawasaki syndrome with intravenous gamma globulin. New England Journal of Medicine 1986;315(6):341–7. - PubMed
Newburger 1991
Newburger 2004
-
- Newburger JW, Takahashi M, Gerber MA, Gewitz MH, Tani LY, Burns JC, et al. Diagnosis, treatment, and long-term management of Kawasaki disease: a statement for health professionals from the Committee on Rheumatic Fever, Endocarditis, and Kawasaki Disease, Council on Cardiovascular Disease in the Young, American Heart Association. Pediatrics 2004;114(6):1708–33. [DOI: 10.1542/peds.2004-2182] - DOI - PubMed
Oates‐Whitehead 2003
Olivieri 2008
-
- Olivieri L, Arling B, Friberg M, Sable C. Coronary artery Z score regression equations and calculators derived from a large heterogeneous population of children undergoing echocardiography. Journal of the American Society of Echocardiography 2009;22(2):159-64. [DOI: 10.1016/j.echo.2008.11.003] [PMID: ] - DOI - PubMed
Onouchi 2008
Onouchi 2010
Onouchi 2012
RevMan Web 2022 [Computer program]
-
- Review Manager Web (RevMan Web). Version 4.12.0. The Cochrane Collaboration, 2022. Available at revman.cochrane.org.
Rife 2020
Rowley 1997
-
- Rowley AH, Eckerley CA, Jäck HM, Shulman ST, Baker SC. IgA plasma cells in vascular tissue of patients with Kawasaki syndrome. Journal of Immunology 1997;159(12):5946–55. - PubMed
Sauvaget 2012
Schünemann 2021
-
- Schünemann HJ, Higgins JP, Vist GE, Glasziou P, Akl EA, Skoetz N, et al. Chapter 14: Completing ‘Summary of findings’ tables and grading the certainty of the evidence. In: Higgins JP, Thomas J, Chandler J, Cumpston M, Li T, Page MJ, Welch VA, editor(s). Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (updated February 2021). Cochrane, 2021. Available from training.cochrane.org/handbook/archive/v6.2.
Tremoulet 2019
Uehara 2003
Verdoni 2020
Yamaji 2019
Yashiro 2004
-
- Yashiro M, Nakamura Y, Yanagawa H. Clinical features of patients with Kawasaki disease whose parents had the same disease. Archives of Pediatrics and Adolescent Medicine 2004;158:1166–9. - PubMed
References to other published versions of this review
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

