Pharmacokinetic/Pharmacodynamic Index Linked to In Vivo Efficacy of the Ampicillin-Ceftriaxone Combination against Enterococcus faecalis
- PMID: 36695584
- PMCID: PMC9933695
- DOI: 10.1128/aac.00966-22
Pharmacokinetic/Pharmacodynamic Index Linked to In Vivo Efficacy of the Ampicillin-Ceftriaxone Combination against Enterococcus faecalis
Abstract
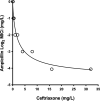
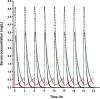
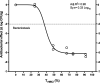
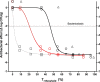
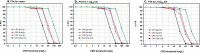
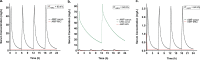
Combination therapy with ampicillin plus ceftriaxone (AMP+CRO) is the first-line therapy for treating severe infections due to Enterococcus faecalis. However, the pharmacokinetic/pharmacodynamic (PK/PD) index linked to the in vivo efficacy of the combination is not yet defined, hindering dose optimization in the clinic. Because classical PK/PD indices are not directly applicable to antimicrobial combinations, two novel indices were tested in the optimized murine model of infection by E. faecalis to delineate the potentiation of AMP by CRO: the time above the CRO threshold (T>threshold) and the time above the AMP instantaneous MIC (T>MICi). The potential clinical relevance was evaluated by simulating human doses of AMP and CRO. Hill's equation fitted well the exposure-response data in terms of T>threshold, with a CRO threshold of 1 mg/L. The required exposures were 46%, 49%, and 52% for stasis and 1- and 2-log10 killing, respectively. Human ceftriaxone doses of 2 g every 12 h (q12h) would reach the target in >90% of strains with thresholds ≤64 mg/L. The AMP T>MICi index also fitted well, and the required exposures were 37%, 41%, and 46% for stasis and 1- and 2-log10 killing, respectively. In humans, the addition of CRO would allow use of lower AMP doses to reach the same T>MICi and to treat strains with higher MICs. This is the first report of the PK/PD indices and required magnitudes linked to AMP+CRO against E. faecalis; these results can be used as the basis to guide the design of clinical trials to improve combined therapy against enterococci.
Keywords: Enterococcus; PK/PD index; antibiotic combination; antimicrobial combinations; pharmacodynamics; pharmacokinetics; synergism.
Conflict of interest statement
The authors declare no conflict of interest.
Figures
Similar articles
-
A new pharmacodynamic approach to study antibiotic combinations against enterococci in vivo: Application to ampicillin plus ceftriaxone.PLoS One. 2020 Dec 8;15(12):e0243365. doi: 10.1371/journal.pone.0243365. eCollection 2020. PLoS One. 2020. PMID: 33290425 Free PMC article.
-
Pharmacodynamics of Ceftaroline plus Ampicillin against Enterococcus faecalis in an In Vitro Pharmacokinetic/Pharmacodynamic Model of Simulated Endocardial Vegetations.Antimicrob Agents Chemother. 2017 Mar 24;61(4):e02235-16. doi: 10.1128/AAC.02235-16. Print 2017 Apr. Antimicrob Agents Chemother. 2017. PMID: 28096164 Free PMC article.
-
Efficacy of Telavancin Alone and in Combination with Ampicillin in a Rat Model of Enterococcus faecalis Endocarditis.Antimicrob Agents Chemother. 2017 May 24;61(6):e02489-16. doi: 10.1128/AAC.02489-16. Print 2017 Jun. Antimicrob Agents Chemother. 2017. PMID: 28320712 Free PMC article.
-
Listeria monocytogenes endocarditis: case report, review of the literature, and laboratory evaluation of potential novel antibiotic synergies.Int J Antimicrob Agents. 2018 Mar;51(3):468-478. doi: 10.1016/j.ijantimicag.2017.12.032. Epub 2018 Jan 11. Int J Antimicrob Agents. 2018. PMID: 29337066 Free PMC article. Review.
-
A Review of Combination Antimicrobial Therapy for Enterococcus faecalis Bloodstream Infections and Infective Endocarditis.Clin Infect Dis. 2018 Jul 2;67(2):303-309. doi: 10.1093/cid/ciy064. Clin Infect Dis. 2018. PMID: 29390132 Free PMC article. Review.
Cited by
-
Treatment of Enterococcus faecalis Infective Endocarditis: A Continuing Challenge.Antibiotics (Basel). 2023 Apr 4;12(4):704. doi: 10.3390/antibiotics12040704. Antibiotics (Basel). 2023. PMID: 37107066 Free PMC article. Review.
-
Pharmacokinetics-Pharmacodynamics Modeling for Evaluating Drug-Drug Interactions in Polypharmacy: Development and Challenges.Clin Pharmacokinet. 2024 Jul;63(7):919-944. doi: 10.1007/s40262-024-01391-2. Epub 2024 Jun 18. Clin Pharmacokinet. 2024. PMID: 38888813 Review.
-
High-Dose Ceftriaxone in Elderly Patients with Enterococcal Infective Endocarditis: Population Pharmacokinetics of Free Ceftriaxone and Dose Optimization.Antibiotics (Basel). 2025 May 15;14(5):508. doi: 10.3390/antibiotics14050508. Antibiotics (Basel). 2025. PMID: 40426574 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

