Synthesis, structural characterization and study of antioxidant and anti-PrPSc properties of flavonoids and their rhenium(I)-tricarbonyl complexes
- PMID: 36695886
- PMCID: PMC9981504
- DOI: 10.1007/s00775-022-01986-9
Synthesis, structural characterization and study of antioxidant and anti-PrPSc properties of flavonoids and their rhenium(I)-tricarbonyl complexes
Abstract
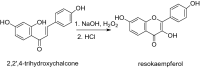
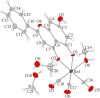
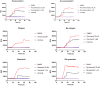
This study aims at the synthesis and initial biological evaluation of novel rhenium-tricarbonyl complexes of 3,3',4',5,7-pentahydroxyflavone (quercetin), 3,7,4΄-trihydroxyflavone (resokaempferol), 5,7-dihydroxyflavone (chrysin) and 4΄,5,7-trihydroxyflavonone (naringenin) as neuroprotective and anti-PrP agents. Resokaempferol was synthesized from 2,2΄,4-trihydroxychalcone by H2O2/NaOH. The rhenium-tricarbonyl complexes of the type fac-[Re(CO)3(Fl)(sol)] were synthesized by reacting the precursor fac-[Re(CO)3(sol)3]+ with an equimolar amount of the flavonoids (Fl) quercetin, resokaempferol, chrysin and naringenin and the solvent (sol) was methanol or water. The respective Re-flavonoid complexes were purified by semi-preparative HPLC and characterized by spectroscopic methods. Furthermore, the structure of Re-chrysin was elucidated by X-ray crystallography. Initial screening of the neuroprotective properties of these compounds included the in vitro assessment of the antioxidant properties by the DPPH assay as well as the anti-lipid peroxidation of linoleic acid in the presence of AAPH and their ability to inhibit soybean lipoxygenase. From the above studies, it was concluded that the complexes' properties are mainly correlated with the structural characteristics and the presence of the flavonoids. The flavonoids and their respective Re-complexes were also tested in vitro for their ability to inhibit the formation and aggregation of the amyloid-like abnormal prion protein, PrPSc, by employing the real-time quaking-induced conversion assay with recombinant PrP seeded with cerebrospinal fluid from patients with Creutzfeldt-Jakob disease. All the compounds blocked de novo abnormal PrP formation and aggregation.
Keywords: Anti-oxidant; Flavonoids; Lipid peroxidation; Lipoxygenase; Prion diseases; Rhenium complexes.
© 2023. The Author(s).
Conflict of interest statement
The authors have no relevant financial or non-financial interests to disclose. No funding was received for conducting this study.
Figures
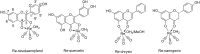
Similar articles
-
Chemical and biological characterization of technetium(I) and Rhenium(I) tricarbonyl complexes with dithioether ligands serving as linkers for coupling the Tc(CO)(3) and Re(CO)(3) moieties to biologically active molecules.Bioconjug Chem. 2000 May-Jun;11(3):414-24. doi: 10.1021/bc990162o. Bioconjug Chem. 2000. PMID: 10821659
-
Guanine and plasmid DNA binding of mono- and trinuclear fac-[Re(CO)3]+ complexes with amino acid ligands.Chembiochem. 2005 Aug;6(8):1397-405. doi: 10.1002/cbic.200400453. Chembiochem. 2005. PMID: 15959921
-
Synthesis and evaluation of new mixed "2 + 1" Re, 99mTc and 186Re tricarbonyl dithiocarbamate complexes with different monodentate ligands.Bioorg Med Chem. 2021 Oct 1;47:116373. doi: 10.1016/j.bmc.2021.116373. Epub 2021 Aug 19. Bioorg Med Chem. 2021. PMID: 34467870
-
Recent development of luminescent rhenium(i) tricarbonyl polypyridine complexes as cellular imaging reagents, anticancer drugs, and antibacterial agents.Dalton Trans. 2017 Dec 21;46(47):16357-16380. doi: 10.1039/c7dt03465b. Epub 2017 Nov 7. Dalton Trans. 2017. PMID: 29110007 Review.
-
Anticancer and Antibiotic Rhenium Tri- and Dicarbonyl Complexes: Current Research and Future Perspectives.Molecules. 2022 Jan 15;27(2):539. doi: 10.3390/molecules27020539. Molecules. 2022. PMID: 35056856 Free PMC article. Review.
Cited by
-
Biomaterials in Cancer Therapy: Investigating the Interaction between Kaempferol and Zinc Ions through Computational, Spectroscopic and Biological Analyses.Materials (Basel). 2024 May 24;17(11):2526. doi: 10.3390/ma17112526. Materials (Basel). 2024. PMID: 38893790 Free PMC article.
-
Gut microbiota and metabolome in sporadic Creutzfeldt-Jakob disease.J Neurol. 2023 Dec;270(12):6021-6032. doi: 10.1007/s00415-023-11961-2. Epub 2023 Aug 29. J Neurol. 2023. PMID: 37642736
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Research Materials

