CD28-CAR-T cell activation through FYN kinase signaling rather than LCK enhances therapeutic performance
- PMID: 36696897
- PMCID: PMC9975250
- DOI: 10.1016/j.xcrm.2023.100917
CD28-CAR-T cell activation through FYN kinase signaling rather than LCK enhances therapeutic performance
Abstract
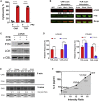
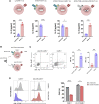
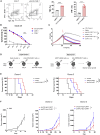
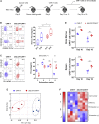
Signal transduction induced by chimeric antigen receptors (CARs) is generally believed to rely on the activity of the SRC family kinase (SFK) LCK, as is the case with T cell receptor (TCR) signaling. Here, we show that CAR signaling occurs in the absence of LCK. This LCK-independent signaling requires the related SFK FYN and a CD28 intracellular domain within the CAR. LCK-deficient CAR-T cells are strongly signaled through CAR and have better in vivo efficacy with reduced exhaustion phenotype and enhanced induction of memory and proliferation. These distinctions can be attributed to the fact that FYN signaling tends to promote proliferation and survival, whereas LCK signaling promotes strong signaling that tends to lead to exhaustion. This non-canonical signaling of CAR-T cells provides insight into the initiation of both TCR and CAR signaling and has important clinical implications for improvement of CAR function.
Keywords: CAR; CAR-T; CD28; FYN; LCK independent signaling; T cell receptor; allogeneic CAR-T; chimeric antigen receptor T cell; co-stimulation; signal transduction; specificity.
Copyright © 2023 The Authors. Published by Elsevier Inc. All rights reserved.
Conflict of interest statement
Declaration of interests National University of Singapore has filed patents based on these findings. P.A.M. is a shareholder and advisory board member of Gen Y Biologics Pte., Ltd. (company registration number: 202005553Z). Patent title: Engineered Immune Cells (PCT/SG2020/050090). Published September 10, 2020: WO 2020/180243 (L.W., N.R.J.G., and J.B.).
Figures




Similar articles
-
Differential T-cell antigen receptor signaling mediated by the Src family kinases Lck and Fyn.Mol Cell Biol. 2000 Feb;20(4):1426-35. doi: 10.1128/MCB.20.4.1426-1435.2000. Mol Cell Biol. 2000. PMID: 10648627 Free PMC article.
-
Tyrosine phosphorylation of Pyk2 is selectively regulated by Fyn during TCR signaling.J Exp Med. 1997 Apr 7;185(7):1253-9. doi: 10.1084/jem.185.7.1253. J Exp Med. 1997. PMID: 9104812 Free PMC article.
-
[Function of the Lck and Fyn in T cell development].Yi Chuan. 2012 Mar;34(3):289-95. doi: 10.3724/sp.j.1005.2012.00289. Yi Chuan. 2012. PMID: 22425947 Review. Chinese.
-
T cell receptor signal initiation induced by low-grade stimulation requires the cooperation of LAT in human T cells.PLoS One. 2010 Nov 30;5(11):e15114. doi: 10.1371/journal.pone.0015114. PLoS One. 2010. PMID: 21152094 Free PMC article.
-
Function of the Src-family kinases, Lck and Fyn, in T-cell development and activation.Oncogene. 2004 Oct 18;23(48):7990-8000. doi: 10.1038/sj.onc.1208074. Oncogene. 2004. PMID: 15489916 Review.
Cited by
-
CAR-T cell therapy for hepatocellular carcinoma: current trends and challenges.Front Immunol. 2024 Nov 6;15:1489649. doi: 10.3389/fimmu.2024.1489649. eCollection 2024. Front Immunol. 2024. PMID: 39569202 Free PMC article. Review.
-
UNC119 regulates T-cell receptor signalling in primary T cells and T acute lymphocytic leukaemia.Life Sci Alliance. 2025 Jan 15;8(3):e202403066. doi: 10.26508/lsa.202403066. Print 2025 Mar. Life Sci Alliance. 2025. PMID: 39814552 Free PMC article.
-
IL-2-inducible T cell kinase deficiency sustains chimeric antigen receptor T cell therapy against tumor cells.J Clin Invest. 2024 Nov 26;135(4):e178558. doi: 10.1172/JCI178558. J Clin Invest. 2024. PMID: 39589809 Free PMC article.
-
Design and development of dual targeting CAR protein for the development of CAR T-cell therapy against KRAS mutated pancreatic ductal adenocarcinoma using computational approaches.Discov Oncol. 2024 Oct 25;15(1):592. doi: 10.1007/s12672-024-01455-6. Discov Oncol. 2024. PMID: 39453574 Free PMC article.
-
Novel FRET-based Immunological Synapse Biosensor for the Prediction of Chimeric Antigen Receptor-T Cell Function.Small Methods. 2025 Mar;9(3):e2401016. doi: 10.1002/smtd.202401016. Epub 2024 Sep 11. Small Methods. 2025. PMID: 39258379 Free PMC article.
References
-
- Kochenderfer J.N., Yu Z., Frasheri D., Restifo N.P., Rosenberg S.A. Adoptive transfer of syngeneic T cells transduced with a chimeric antigen receptor that recognizes murine CD19 can eradicate lymphoma and normal B cells. Blood. 2010;116:3875–3886. doi: 10.1182/blood-2010-01-265041. - DOI - PMC - PubMed
-
- Salter A.I., Ivey R.G., Kennedy J.J., Voillet V., Rajan A., Alderman E.J., Voytovich U.J., Lin C., Sommermeyer D., Liu L., et al. Phosphoproteomic analysis of chimeric antigen receptor signaling reveals kinetic and quantitative differences that affect cell function. Sci. Signal. 2018;11:eaat6753. doi: 10.1126/scisignal.aat6753. - DOI - PMC - PubMed
-
- Davenport A.J., Cross R.S., Watson K.A., Liao Y., Shi W., Prince H.M., Beavis P.A., Trapani J.A., Kershaw M.H., Ritchie D.S., et al. Chimeric antigen receptor T cells form nonclassical and potent immune synapses driving rapid cytotoxicity. Proc. Natl. Acad. Sci. USA. 2018;115:E2068–E2076. doi: 10.1073/pnas.1716266115. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

