Activation of L-lactate oxidase by the formation of enzyme assemblies through liquid-liquid phase separation
- PMID: 36697449
- PMCID: PMC9877012
- DOI: 10.1038/s41598-023-28040-1
Activation of L-lactate oxidase by the formation of enzyme assemblies through liquid-liquid phase separation
Abstract
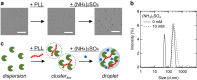
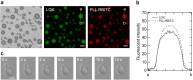
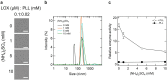
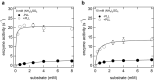
The assembly state of enzymes is gaining interest as a mechanism for regulating the function of enzymes in living cells. One of the current topics in enzymology is the relationship between enzyme activity and the assembly state due to liquid-liquid phase separation. In this study, we demonstrated enzyme activation via the formation of enzyme assemblies using L-lactate oxidase (LOX). LOX formed hundreds of nanometer-scale assemblies with poly-L-lysine (PLL). In the presence of ammonium sulfate, the LOX-PLL clusters formed micrometer-scale liquid droplets. The enzyme activities of LOX in clusters and droplets were one order of magnitude higher than those in the dispersed state, owing to a decrease in KM and an increase in kcat. Moreover, the clusters exhibited a higher activation effect than the droplets. In addition, the conformation of LOX changed in the clusters, resulting in increased enzyme activation. Understanding enzyme activation and assembly states provides important information regarding enzyme function in living cells, in addition to biotechnology applications.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

