A platinum(IV) prodrug strategy to overcome glutathione-based oxaliplatin resistance
- PMID: 36697790
- PMCID: PMC9814792
- DOI: 10.1038/s42004-022-00661-z
A platinum(IV) prodrug strategy to overcome glutathione-based oxaliplatin resistance
Abstract
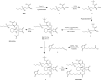
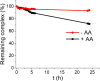
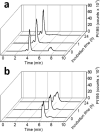
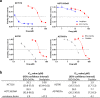
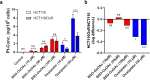
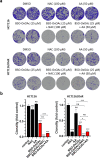
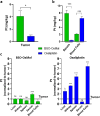
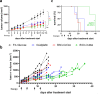
Clinical efficacy of oxaliplatin is frequently limited by severe adverse effects and therapy resistance. Acquired insensitivity to oxaliplatin is, at least in part, associated with elevated levels of glutathione (GSH). In this study we report on an oxaliplatin-based platinum(IV) prodrug, which releases L-buthionine-S,R-sulfoximine (BSO), an inhibitor of glutamate-cysteine ligase, the rate-limiting enzyme in GSH biosynthesis. Two complexes bearing either acetate (BSO-OxOAc) or an albumin-binding maleimide (BSO-OxMal) as second axial ligand were synthesized and characterized. The in vitro anticancer activity of BSO-OxOAc was massively reduced in comparison to oxaliplatin, proving its prodrug nature. Nevertheless, the markedly lower intracellular oxaliplatin uptake in resistant HCT116/OxR cells was widely overcome by BSO-OxOAc resulting in distinctly reduced resistance levels. Platinum accumulation in organs of a colorectal cancer mouse model revealed higher tumor selectivity of BSO-OxMal as compared to oxaliplatin. This corresponded with increased antitumor activity, resulting in significantly enhanced overall survival. BSO-OxMal-treated tumors exhibited reduced GSH levels, proliferative activity and enhanced DNA damage (pH2AX) compared to oxaliplatin. Conversely, pH2AX staining especially in kidney cells was distinctly increased by oxaliplatin but not by BSO-OxMal. Taken together, our data provide compelling evidence for enhanced tumor specificity of the oxaliplatin(IV)/BSO prodrug.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interest.
Figures
Similar articles
-
d,l-buthionine-(S,R)-sulfoximine potentiates in vivo the therapeutic efficacy of doxorubicin against multidrug resistance protein-expressing tumors.Clin Cancer Res. 1996 Dec;2(12):1961-8. Clin Cancer Res. 1996. PMID: 9816155
-
Albumin-targeting of an oxaliplatin-releasing platinum(iv) prodrug results in pronounced anticancer activity due to endocytotic drug uptake in vivo.Chem Sci. 2021 Aug 26;12(38):12587-12599. doi: 10.1039/d1sc03311e. eCollection 2021 Oct 6. Chem Sci. 2021. PMID: 34703544 Free PMC article.
-
Effect of buthionine sulfoximine on PtII and PtIV drug accumulation and the formation of glutathione conjugates in human ovarian-carcinoma cell lines.Int J Cancer. 1993 Nov 11;55(5):848-56. doi: 10.1002/ijc.2910550526. Int J Cancer. 1993. PMID: 8244583
-
Enhanced melphalan cytotoxicity in human ovarian cancer in vitro and in tumor-bearing nude mice by buthionine sulfoximine depletion of glutathione.Biochem Pharmacol. 1987 Jan 1;36(1):147-53. doi: 10.1016/0006-2952(87)90392-3. Biochem Pharmacol. 1987. PMID: 3801051
-
Promotion of trans-platinum in vivo effects on renal heme and hemoprotein metabolism by D,L-buthionine-S,R-sulfoximine. Possible role of glutathione.Biochem Pharmacol. 1990 May 15;39(10):1565-71. doi: 10.1016/0006-2952(90)90522-m. Biochem Pharmacol. 1990. PMID: 2337413
Cited by
-
Dual acting oxaliplatin (IV) prodrug loaded albumin nanoparticles for safer synergistic anticancer action against triple negative breast cancer.Drug Deliv Transl Res. 2025 Mar 11. doi: 10.1007/s13346-025-01833-9. Online ahead of print. Drug Deliv Transl Res. 2025. PMID: 40069528
-
Albumin-targeted oxaliplatin(iv) prodrugs bearing STING agonists.Inorg Chem Front. 2025 Apr 4;12(13):4284-4305. doi: 10.1039/d5qi00433k. eCollection 2025 Jun 24. Inorg Chem Front. 2025. PMID: 40191696 Free PMC article.
-
Combination of Drug Delivery Properties of PAMAM Dendrimers and Cytotoxicity of Platinum(IV) Complexes-A More Selective Anticancer Treatment?Pharmaceutics. 2023 May 17;15(5):1515. doi: 10.3390/pharmaceutics15051515. Pharmaceutics. 2023. PMID: 37242758 Free PMC article.
-
Chemotherapeutic Potential of Chlorambucil-Platinum(IV) Prodrugs against Cisplatin-Resistant Colorectal Cancer Cells.Int J Mol Sci. 2024 Jul 28;25(15):8252. doi: 10.3390/ijms25158252. Int J Mol Sci. 2024. PMID: 39125821 Free PMC article.
-
AKR1C1 interacts with STAT3 to increase intracellular glutathione and confers resistance to oxaliplatin in colorectal cancer.Acta Pharm Sin B. 2024 Dec;14(12):5305-5320. doi: 10.1016/j.apsb.2024.08.031. Epub 2024 Sep 2. Acta Pharm Sin B. 2024. PMID: 39807317 Free PMC article.
References
-
- WHO. Cancer, <https://www.who.int/news-room/fact-sheets/detail/cancer> (2020).
-
- WHO. Cancer Today, <https://gco.iarc.fr/today/fact-sheets-cancers> (2021).
-
- Deo KM, et al. Platinum coordination compounds with potent anticancer activity. Coord. Chem. Rev. 2018;375:148–163. doi: 10.1016/j.ccr.2017.11.014. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

