Switchable aqueous catalytic systems for organic transformations
- PMID: 36697818
- PMCID: PMC9814960
- DOI: 10.1038/s42004-022-00734-z
Switchable aqueous catalytic systems for organic transformations
Abstract
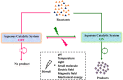
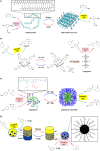
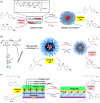
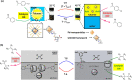
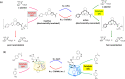
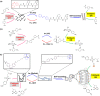
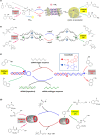
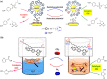
In living organisms, enzyme catalysis takes place in aqueous media with extraordinary spatiotemporal control and precision. The mechanistic knowledge of enzyme catalysis and related approaches of creating a suitable microenvironment for efficient chemical transformations have been an important source of inspiration for the design of biomimetic artificial catalysts. However, in "nature-like" environments, it has proven difficult for artificial catalysts to promote effective chemical transformations. Besides, control over reaction rate and selectivity are important for smart application purposes. These can be achieved via incorporation of stimuli-responsive features into the structure of smart catalytic systems. Here, we summarize such catalytic systems whose activity can be switched 'on' or 'off' by the application of stimuli in aqueous environments. We describe the switchable catalytic systems capable of performing organic transformations with classification in accordance to the stimulating agent. Switchable catalytic activity in aqueous environments provides new possibilities for the development of smart materials for biomedicine and chemical biology. Moreover, engineering of aqueous catalytic systems can be expected to grow in the coming years with a further broadening of its application to diverse fields.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures

References
Publication types
Grants and funding
LinkOut - more resources
Full Text Sources

