Nickel-catalyzed alkyl-arylation of 3,3,3-trifluoropropene
- PMID: 36697891
- PMCID: PMC9814099
- DOI: 10.1038/s42004-022-00659-7
Nickel-catalyzed alkyl-arylation of 3,3,3-trifluoropropene
Abstract
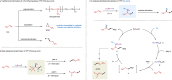
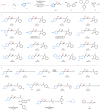
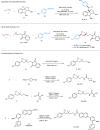
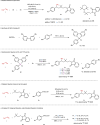
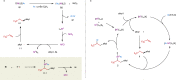
Owing to the versatile synthetic utility of its carbon-carbon double bond, low-cost industrial chemical 3,3,3-trifluoropropene (TFP) represents one of the most straightforward and cost-efficient precursors to prepare trifluoromethylated compounds. However, only limited methods for the efficient transformations of TFP have been reported so far. Here, we report a nickel-catalyzed dicarbofunctionalization of TFP. The reaction uses inexpensive NiCl2·6H2O as the catalyst and 4,4'-biMeO-bpy and PCy2Ph as the ligands, allowing the alkyl-arylation of TFP with a variety of tertiary alkyl iodides and arylzinc reagents in high efficiency. This nickel-catalyzed process overcomes the previous challenges by suppressing β-H and β-F eliminations from TFP, rendering this strategy effective for the transformations of TFP into medicinal interest trifluoromethylated compounds.
© 2022. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Enantioselective nickel-catalyzed dicarbofunctionalization of 3,3,3-trifluoropropene.Nat Commun. 2022 Sep 21;13(1):5539. doi: 10.1038/s41467-022-33159-2. Nat Commun. 2022. PMID: 36130927 Free PMC article.
-
Transition-Metal (Pd, Ni, Mn)-Catalyzed C-C Bond Constructions Involving Unactivated Alkyl Halides and Fundamental Synthetic Building Blocks.Acc Chem Res. 2019 Apr 16;52(4):1134-1144. doi: 10.1021/acs.accounts.9b00044. Epub 2019 Mar 25. Acc Chem Res. 2019. PMID: 30908013 Free PMC article.
-
Nickel-Catalyzed Stereoselective Coupling Reactions of Benzylic and Alkyl Alcohol Derivatives.Acc Chem Res. 2023 Nov 21;56(22):3313-3324. doi: 10.1021/acs.accounts.3c00547. Epub 2023 Nov 7. Acc Chem Res. 2023. PMID: 37936256 Free PMC article.
-
Three-component 1,2-dicarbofunctionalization of alkenes involving alkyl radicals.Chem Commun (Camb). 2022 Jan 18;58(6):730-746. doi: 10.1039/d1cc05730h. Chem Commun (Camb). 2022. PMID: 34931629 Review.
-
Ni-Catalyzed C-C Couplings Using Alkyl Electrophiles.Top Curr Chem (Cham). 2016 Oct;374(5):66. doi: 10.1007/s41061-016-0067-6. Epub 2016 Aug 31. Top Curr Chem (Cham). 2016. PMID: 27580894 Review.
Cited by
-
Nickel-Catalyzed Regioselective Intermolecular Dialkylation of Alkenylarenes: Generation of Two Vicinal C(sp3 )-C(sp3 ) Bonds Across Alkenes.Angew Chem Int Ed Engl. 2023 Aug 7;62(32):e202305522. doi: 10.1002/anie.202305522. Epub 2023 Jun 29. Angew Chem Int Ed Engl. 2023. PMID: 37316459 Free PMC article.
-
Iron-Mediated Dialkylation of Alkenylarenes with Benzyl Bromides.J Org Chem. 2024 Nov 15;89(22):16292-16299. doi: 10.1021/acs.joc.3c02548. Epub 2024 Apr 4. J Org Chem. 2024. PMID: 38572911
References
-
- Goldschmidt A. The Polymerization of 3,3,3-Trifluoropropene and 2-Methyl-3,3,3-trifluoropropene. J. Am. Chem. Soc. 1951;73:2940. doi: 10.1021/ja01150a508. - DOI
-
- Kostov G, Ameduri B, Brandstadter SM. Radical telomerization of 3,3,3-trifluoropropene with diethyl hydrogen phosphonate: Characterization of the first telomeric adducts and assessment of the transfer constants. J. Fluor. Chem. 2007;128:910–918. doi: 10.1016/j.jfluchem.2007.03.015. - DOI
-
- Kostov GK, Améduri B, Brandstadter S. Telomerization of 3,3,3-Trifluoroprop-1-ene and Functionalization of Its Telomers. Collect. Czech. Chem. Commun. 2008;73:1747–1763. doi: 10.1135/cccc20081747. - DOI
-
- Bruno A. Controlled Radical (Co)polymerization of Fluoromonomers. Macromolecules. 2010;43:10163–10184. doi: 10.1021/ma1019297. - DOI
-
- Soulestin T, Ladmiral V, Dos Santos FD, Améduri B. Vinylidene fluoride- and trifluoroethylene-containing fluorinated electroactive copolymers. How does chemistry impact properties? Prog. Polym. Sci. 2017;72:16–60. doi: 10.1016/j.progpolymsci.2017.04.004. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

