Anti-inflammatory effects of β-FNA are sex-dependent in a pre-clinical model of LPS-induced inflammation
- PMID: 36698151
- PMCID: PMC9878921
- DOI: 10.1186/s12950-023-00328-z
Anti-inflammatory effects of β-FNA are sex-dependent in a pre-clinical model of LPS-induced inflammation
Abstract
Background: Inflammation is present in neurological and peripheral disorders. Thus, targeting inflammation has emerged as a viable option for treating these disorders. Previous work indicated pretreatment with beta-funaltrexamine (β-FNA), a selective mu-opioid receptor (MOR) antagonist, inhibited inflammatory signaling in vitro in human astroglial cells, as well as lipopolysaccharide (LPS)-induced neuroinflammation and sickness-like-behavior in mice. This study explores the protective effects of β-FNA when treatment occurs 10 h after LPS administration and is the first-ever investigation of the sex-dependent effects of β-FNA on LPS-induced inflammation in the brain and peripheral tissues, including the intestines.
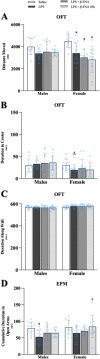
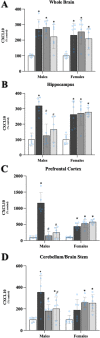
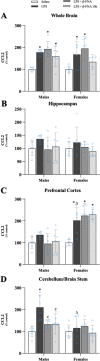
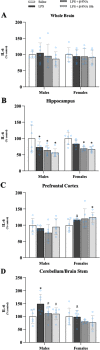
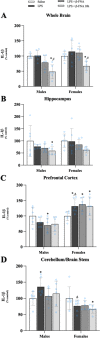
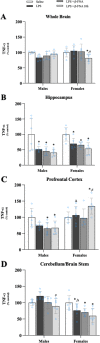
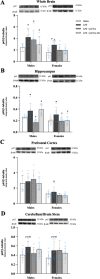
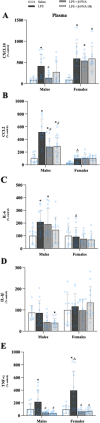
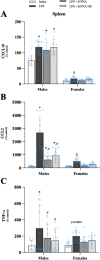
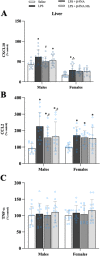
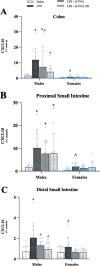
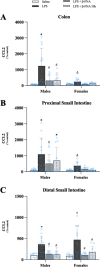
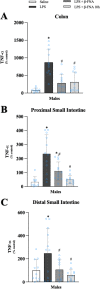
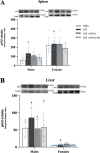
Results: Male and female C57BL/6J mice were administered LPS followed by treatment with β-FNA-immediately or 10 h post-LPS. Sickness- and anxiety-like behavior were assessed using an open-field test and an elevated-plus-maze test, followed by the collection of whole brain, hippocampus, prefrontal cortex, cerebellum/brain stem, plasma, spleen, liver, large intestine (colon), proximal small intestine, and distal small intestine. Levels of inflammatory chemokines/cytokines (interferon γ-induced-protein, IP-10 (CXCL10); monocyte-chemotactic-protein 1, MCP-1 (CCL2); interleukin-6, IL-6; interleukin-1β, IL-1β; and tumor necrosis factor-alpha, TNF-α) in tissues were measured using an enzyme-linked immunosorbent assay. Western blot analysis was used to assess nuclear factor-kappa B (NF-κB) expression. There were sex-dependent differences in LPS-induced inflammation across brain regions and peripheral tissues. Overall, LPS-induced CXCL10, CCL2, TNF-α, and NF-κB were most effectively downregulated by β-FNA; and β-FNA effects differed across brain regions, peripheral tissues, timing of the dose, and in some instances, in a sex-dependent manner. β-FNA reduced LPS-induced anxiety-like behavior most effectively in female mice.
Conclusion: These findings provide novel insights into the sex-dependent anti-inflammatory effects of β-FNA and advance this agent as a potential therapeutic option for reducing both neuroinflammation an intestinal inflammation.
Keywords: Chemokine; Cytokine; Neuroinflammation; Neuroprotective; Nuclear factor-κB; Opioid; Peripheral inflammation; β-funaltrexamine.
© 2023. The Author(s).
Conflict of interest statement
None.
Figures
Similar articles
-
The effects of chronic, continuous β-funaltrexamine pre-treatment on lipopolysaccharide-induced inflammation and behavioral deficits in C57BL/6J mice.J Inflamm (Lond). 2024 Sep 2;21(1):33. doi: 10.1186/s12950-024-00407-9. J Inflamm (Lond). 2024. PMID: 39223594 Free PMC article.
-
Anti-inflammatory actions of β-funaltrexamine in a mouse model of lipopolysaccharide-induced inflammation.Inflammopharmacology. 2023 Feb;31(1):349-358. doi: 10.1007/s10787-022-01113-9. Epub 2022 Dec 17. Inflammopharmacology. 2023. PMID: 36527567
-
The opioid antagonist, β-funaltrexamine, inhibits lipopolysaccharide-induced neuroinflammation and reduces sickness behavior in mice.Physiol Behav. 2017 May 1;173:52-60. doi: 10.1016/j.physbeh.2017.01.037. Epub 2017 Jan 24. Physiol Behav. 2017. PMID: 28130086
-
The opioid antagonist, β-funaltrexamine, inhibits NF-κB signaling and chemokine expression in human astrocytes and in mice.Eur J Pharmacol. 2015 Sep 5;762:193-201. doi: 10.1016/j.ejphar.2015.05.040. Epub 2015 May 22. Eur J Pharmacol. 2015. PMID: 26007645 Free PMC article.
-
The opioid antagonist, beta-funaltrexamine, inhibits chemokine expression in human astroglial cells.J Neuroimmunol. 2007 May;186(1-2):141-9. doi: 10.1016/j.jneuroim.2007.03.021. Epub 2007 May 1. J Neuroimmunol. 2007. PMID: 17475341 Free PMC article.
Cited by
-
The effects of chronic, continuous β-funaltrexamine pre-treatment on lipopolysaccharide-induced inflammation and behavioral deficits in C57BL/6J mice.J Inflamm (Lond). 2024 Sep 2;21(1):33. doi: 10.1186/s12950-024-00407-9. J Inflamm (Lond). 2024. PMID: 39223594 Free PMC article.
References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

