Wireless Self-Powered Optogenetic System for Long-Term Cardiac Neuromodulation to Improve Post-MI Cardiac Remodeling and Malignant Arrhythmia
- PMID: 36698262
- PMCID: PMC10037959
- DOI: 10.1002/advs.202205551
Wireless Self-Powered Optogenetic System for Long-Term Cardiac Neuromodulation to Improve Post-MI Cardiac Remodeling and Malignant Arrhythmia
Abstract
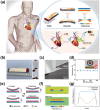
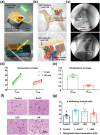
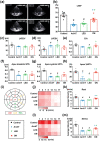
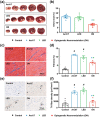
Autonomic imbalance is an important characteristic of patients after myocardial infarction (MI) and adversely contributes to post-MI cardiac remodeling and ventricular arrhythmias (VAs). A previous study proved that optogenetic modulation could precisely inhibit cardiac sympathetic hyperactivity and prevent acute ischemia-induced VAs. Here, a wireless self-powered optogenetic modulation system is introduced, which achieves long-term precise cardiac neuromodulation in ambulatory canines. The wireless self-powered optical system based on a triboelectric nanogenerator is powered by energy harvested from body motion and realized the effective optical illumination that is required for optogenetic neuromodulation (ON). It is further demonstrated that long-term ON significantly mitigates MI-induced sympathetic remodeling and hyperactivity, and improves a variety of clinically relevant outcomes such as improves ventricular dysfunction, reduces infarct size, increases electrophysiological stability, and reduces susceptibility to VAs. These novel insights suggest that wireless ON holds translational potential for the clinical treatment of arrhythmia and other cardiovascular diseases related to sympathetic hyperactivity. Moreover, this innovative self-powered optical system may provide an opportunity to develop implantable/wearable and self-controllable devices for long-term optogenetic therapy.
Keywords: cardiac remodeling; neuromodulation; triboelectric nanogenerator; ventricular arrhythmia; wireless self-powered optogenetics.
© 2023 The Authors. Advanced Science published by Wiley-VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures



Similar articles
-
Sonogenetic neuromodulation prevents post-myocardial infarction malignant arrhythmia and cardiac remodeling through the brain-heart circuit.Basic Res Cardiol. 2025 Jul 9. doi: 10.1007/s00395-025-01130-z. Online ahead of print. Basic Res Cardiol. 2025. PMID: 40632283
-
Optogenetic Modulation of Cardiac Sympathetic Nerve Activity to Prevent Ventricular Arrhythmias.J Am Coll Cardiol. 2017 Dec 5;70(22):2778-2790. doi: 10.1016/j.jacc.2017.09.1107. J Am Coll Cardiol. 2017. PMID: 29191327
-
Metabolism regulator adiponectin prevents cardiac remodeling and ventricular arrhythmias via sympathetic modulation in a myocardial infarction model.Basic Res Cardiol. 2022 Jul 11;117(1):34. doi: 10.1007/s00395-022-00939-2. Basic Res Cardiol. 2022. PMID: 35819552
-
Clinical aspects of left ventricular diastolic function assessed by Doppler echocardiography following acute myocardial infarction.Dan Med Bull. 2001 Nov;48(4):199-210. Dan Med Bull. 2001. PMID: 11767125 Review.
-
Sympathetic nerve sprouting, electrical remodeling and the mechanisms of sudden cardiac death.Cardiovasc Res. 2001 May;50(2):409-16. doi: 10.1016/s0008-6363(00)00308-4. Cardiovasc Res. 2001. PMID: 11334845 Review.
Cited by
-
The proarrhythmogenic role of autonomics and emerging neuromodulation approaches to prevent sudden death in cardiac ion channelopathies.Cardiovasc Res. 2024 Mar 13;120(2):114-131. doi: 10.1093/cvr/cvae009. Cardiovasc Res. 2024. PMID: 38195920 Free PMC article. Review.
-
Emerging optogenetics technologies in biomedical applications.Smart Med. 2023 Nov 1;2(4):e20230026. doi: 10.1002/SMMD.20230026. eCollection 2023 Nov. Smart Med. 2023. PMID: 39188295 Free PMC article. Review.
-
Advances in Biointegrated Wearable and Implantable Optoelectronic Devices for Cardiac Healthcare.Cyborg Bionic Syst. 2024 Oct 18;5:0172. doi: 10.34133/cbsystems.0172. eCollection 2024. Cyborg Bionic Syst. 2024. PMID: 39431246 Free PMC article. Review.
-
Ultra-low frequency magnetic energy focusing for highly effective wireless powering of deep-tissue implantable electronic devices.Natl Sci Rev. 2024 Feb 28;11(5):nwae062. doi: 10.1093/nsr/nwae062. eCollection 2024 May. Natl Sci Rev. 2024. PMID: 38628571 Free PMC article.
-
Novel Electroactive Therapeutic Platforms for Cardiac Arrhythmia Management.Adv Sci (Weinh). 2025 Jun;12(24):e2500061. doi: 10.1002/advs.202500061. Epub 2025 Feb 14. Adv Sci (Weinh). 2025. PMID: 39951007 Free PMC article. Review.
References
-
- Shen M. J., Zipes D. P., Circ. Res. 2014, 114, 1004. - PubMed
-
- Zhou S., Jung B. C., Tan A. Y., Trang V. Q., Gholmieh G., Han S. W., Lin S. F., Fishbein M. C., Chen P. S., Chen L. S., Heart Rhythm 2008, 5, 131. - PubMed
-
- Cao J. M., Chen L. S., KenKnight B. H., Ohara T., Lee M. H., Tsai J., Lai W. W., Karagueuzian H. S., Wolf P. L., Fishbein M. C., Chen P. S., Circ. Res. 2000, 86, 816. - PubMed
-
- Odero A., Bozzani A., De Ferrari G. M., Schwartz P. J., Heart Rhythm 2010, 7, 1161. - PubMed
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous
