Non-deacetylated poly- N-acetylglucosamine-hyperproducing Staphylococcus aureus undergoes immediate autoaggregation upon vortexing
- PMID: 36699608
- PMCID: PMC9868172
- DOI: 10.3389/fmicb.2022.1101545
Non-deacetylated poly- N-acetylglucosamine-hyperproducing Staphylococcus aureus undergoes immediate autoaggregation upon vortexing
Abstract
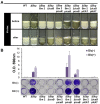
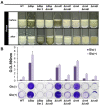
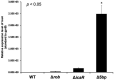
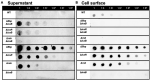
Biofilms are microbial communities of cells embedded in a matrix of extracellular polymeric substances generated and adhering to each other or to a surface. Cell aggregates formed in the absence of a surface and floating pellicles that form biofilms at the air-liquid interface are also considered to be a type of biofilm. Staphylococcus aureus is a well-known cause of biofilm infections and high-molecular-weight polysaccharides, poly-N-acetylglucosamine (PNAG) is a main constituent of the biofilm. An icaADBC operon comprises major machinery to synthesize and extracellularly secrete PNAG. Extracellular PNAG is partially deacetylated by IcaB deacetylase, and the positively charged PNAG hence interacts with negatively charged cell surface to form the major component of biofilm. We previously reported a new regulator of biofilm (Rob) and demonstrated that Rob binds to a unique 5-bp motif, TATTT, present in intergenic region between icaADBC operon and its repressor gene icaR in Yu et al. The deletion of the 5-bp motif induces excessive adherent biofilm formation. The real function of the 5-bp motif is still unknown. In an attempt to isolate the 5-bp motif deletion mutant, we isolated several non-adherent mutants. They grew normally in turbid broth shaking culture but immediately auto-aggregated upon weak vortexing and sedimented as a lump resulting in a clear supernatant. Whole genome sequencing of the mutants identified they all carried mutations in icaB in addition to deletion of the 5-bp motif. Purification and molecular characterization of auto-aggregating factor in the culture supernatant of the mutant identified that the factor was a massively produced non-deacetylated PNAG. Therefore, we created a double deficient strain of biofilm inhibitory factors (5-bp motif, icaR, rob) and icaB to confirm the aggregation phenomenon. This peculiar phenomenon was only observed in Δ5bpΔicaB double mutant but not in ΔicaR ΔicaB or ΔrobΔicaB mutant. This study explains large amount of extracellularly produced non-deacetylated PNAG by Δ5bpΔicaB double mutation induced rapid auto-aggregation of S. aureus cells by vortexing. This phenomenon indicated that Staphylococcus aureus may form biofilms that do not adhere to solid surfaces and we propose this as a new mechanism of non-adherent biofilm formation of S. aureus.
Keywords: PNAG; Staphylococcus aureus; aggregation; biofilm; icaB; poly-N-acetylglucosamine.
Copyright © 2023 Kutsuno, Hayashi, Yu, Yamada, Hisatsune and Sugai.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
A Novel Repressor of the ica Locus Discovered in Clinically Isolated Super-Biofilm-Elaborating Staphylococcus aureus.mBio. 2017 Jan 31;8(1):e02282-16. doi: 10.1128/mBio.02282-16. mBio. 2017. PMID: 28143981 Free PMC article.
-
New Molecular Mechanism of Superbiofilm Elaboration in a Staphylococcus aureus Clinical Strain.Microbiol Spectr. 2023 Jan 31;11(2):e0442522. doi: 10.1128/spectrum.04425-22. Online ahead of print. Microbiol Spectr. 2023. PMID: 36719203 Free PMC article.
-
Rbf promotes biofilm formation by Staphylococcus aureus via repression of icaR, a negative regulator of icaADBC.J Bacteriol. 2009 Oct;191(20):6363-73. doi: 10.1128/JB.00913-09. Epub 2009 Aug 14. J Bacteriol. 2009. PMID: 19684134 Free PMC article.
-
Polysaccharide intercellular adhesin in biofilm: structural and regulatory aspects.Front Cell Infect Microbiol. 2015 Feb 10;5:7. doi: 10.3389/fcimb.2015.00007. eCollection 2015. Front Cell Infect Microbiol. 2015. PMID: 25713785 Free PMC article. Review.
-
Genetic regulation of the intercellular adhesion locus in staphylococci.Front Cell Infect Microbiol. 2012 Mar 26;2:38. doi: 10.3389/fcimb.2012.00038. eCollection 2012. Front Cell Infect Microbiol. 2012. PMID: 23061050 Free PMC article. Review.
Cited by
-
Adaptive traits of Nitrosocosmicus clade ammonia-oxidizing archaea.mBio. 2024 Nov 13;15(11):e0216924. doi: 10.1128/mbio.02169-24. Epub 2024 Oct 3. mBio. 2024. PMID: 39360821 Free PMC article.
References
LinkOut - more resources
Full Text Sources

