Age-related changes in tau and autophagy in human brain in the absence of neurodegeneration
- PMID: 36701399
- PMCID: PMC9879510
- DOI: 10.1371/journal.pone.0262792
Age-related changes in tau and autophagy in human brain in the absence of neurodegeneration
Abstract
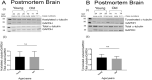
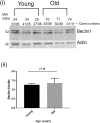
Tau becomes abnormally hyper-phosphorylated and aggregated in tauopathies like Alzheimers disease (AD). As age is the greatest risk factor for developing AD, it is important to understand how tau protein itself, and the pathways implicated in its turnover, change during aging. We investigated age-related changes in total and phosphorylated tau in brain samples from two cohorts of cognitively normal individuals spanning 19-74 years, without overt neurodegeneration. One cohort utilised resected tissue and the other used post-mortem tissue. Total soluble tau levels declined with age in both cohorts. Phosphorylated tau was undetectable in the post-mortem tissue but was clearly evident in the resected tissue and did not undergo significant age-related change. To ascertain if the decline in soluble tau was correlated with age-related changes in autophagy, three markers of autophagy were tested but only two appeared to increase with age and the third was unchanged. This implies that in individuals who do not develop neurodegeneration, there is an age-related reduction in soluble tau which could potentially be due to age-related changes in autophagy. Thus, to explore how an age-related increase in autophagy might influence tau-mediated dysfunctions in vivo, autophagy was enhanced in a Drosophila model and all age-related tau phenotypes were significantly ameliorated. These data shed light on age-related physiological changes in proteins implicated in AD and highlights the need to study pathways that may be responsible for these changes. It also demonstrates the therapeutic potential of interventions that upregulate turnover of aggregate-prone proteins during aging.
Copyright: © 2023 Chatterjee et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures


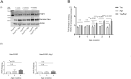
Similar articles
-
Autophagic and lysosomal defects in human tauopathies: analysis of post-mortem brain from patients with familial Alzheimer disease, corticobasal degeneration and progressive supranuclear palsy.Acta Neuropathol Commun. 2016 Mar 2;4:22. doi: 10.1186/s40478-016-0292-9. Acta Neuropathol Commun. 2016. PMID: 26936765 Free PMC article.
-
Reduced expression of dMyc mitigates TauV337M mediated neurotoxicity by preventing the Tau hyperphosphorylation and inducing autophagy in Drosophila.Neurosci Lett. 2020 Jan 10;715:134622. doi: 10.1016/j.neulet.2019.134622. Epub 2019 Nov 9. Neurosci Lett. 2020. PMID: 31715291
-
S6K/p70S6K1 protects against tau-mediated neurodegeneration by decreasing the level of tau phosphorylated at Ser262 in a Drosophila model of tauopathy.Neurobiol Aging. 2018 Nov;71:255-264. doi: 10.1016/j.neurobiolaging.2018.07.021. Epub 2018 Aug 3. Neurobiol Aging. 2018. PMID: 30172839
-
Tau pathology in Alzheimer disease and other tauopathies.Biochim Biophys Acta. 2005 Jan 3;1739(2-3):198-210. doi: 10.1016/j.bbadis.2004.09.008. Biochim Biophys Acta. 2005. PMID: 15615638 Review.
-
Tauopathy: A common mechanism for neurodegeneration and brain aging.Mech Ageing Dev. 2019 Mar;178:72-79. doi: 10.1016/j.mad.2019.01.007. Epub 2019 Jan 19. Mech Ageing Dev. 2019. PMID: 30668956 Free PMC article. Review.
Cited by
-
Cognitive capacity in amyotrophic lateral sclerosis: the value of diagnostic markers in cerebrospinal fluid and the influence of nutrition and pulmonary function.Brain Commun. 2025 Apr 8;7(2):fcaf137. doi: 10.1093/braincomms/fcaf137. eCollection 2025. Brain Commun. 2025. PMID: 40241787 Free PMC article.
-
Autophagy and Alzheimer's Disease: Mechanisms and Impact Beyond the Brain.Cells. 2025 Jun 16;14(12):911. doi: 10.3390/cells14120911. Cells. 2025. PMID: 40558538 Free PMC article. Review.
-
Nuclear face of Tau: an inside player in neurodegeneration.Acta Neuropathol Commun. 2023 Dec 12;11(1):196. doi: 10.1186/s40478-023-01702-x. Acta Neuropathol Commun. 2023. PMID: 38087392 Free PMC article. Review.
-
Impaired biogenesis of basic proteins impacts multiple hallmarks of the aging brain.bioRxiv [Preprint]. 2024 Jan 9:2023.07.20.549210. doi: 10.1101/2023.07.20.549210. bioRxiv. 2024. PMID: 38260253 Free PMC article. Preprint.
-
Aging, NRF2, and TAU: A Perfect Match for Neurodegeneration?Antioxidants (Basel). 2023 Aug 4;12(8):1564. doi: 10.3390/antiox12081564. Antioxidants (Basel). 2023. PMID: 37627559 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases
Research Materials

