COVID-19 severity by vaccination status in the NCI COVID-19 and Cancer Patients Study (NCCAPS)
- PMID: 36702472
- PMCID: PMC10165483
- DOI: 10.1093/jnci/djad015
COVID-19 severity by vaccination status in the NCI COVID-19 and Cancer Patients Study (NCCAPS)
Erratum in
-
Correction to: COVID-19 severity by vaccination status in the NCI COVID-19 and Cancer Patients Study (NCCAPS).J Natl Cancer Inst. 2023 Jun 8;115(6):771-772. doi: 10.1093/jnci/djad076. J Natl Cancer Inst. 2023. PMID: 37178175 Free PMC article. No abstract available.
Abstract
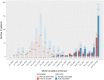
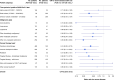
We investigated the association of SARS CoV-2 vaccination with COVID-19 severity in a longitudinal study of adult cancer patients with COVID-19. A total of 1610 patients who were within 14 days of an initial positive SARS CoV-2 test and had received recent anticancer treatment or had a history of stem cell transplant or CAR-T cell therapy were enrolled between May 21, 2020, and February 1, 2022. Patients were considered fully vaccinated if they were 2 weeks past their second dose of mRNA vaccine (BNT162b2 or mRNA-1273) or a single dose of adenovirus vector vaccine (Ad26.COV2.S) at the time of positive SARS CoV-2 test. We defined severe COVID-19 disease as hospitalization for COVID-19 or death within 30 days. Vaccinated patients were significantly less likely to develop severe disease compared with those who were unvaccinated (odds ratio = 0.44, 95% confidence interval = 0.28 to 0.72, P < .001). These results support COVID-19 vaccination among cancer patients receiving active immunosuppressive treatment.
Published by Oxford University Press 2023.
Figures
Comment in
-
RE: COVID-19 severity by vaccination status in the NCI COVID-19 and Cancer Patients Study (NCCAPS).J Natl Cancer Inst. 2023 Jun 8;115(6):766-767. doi: 10.1093/jnci/djad045. J Natl Cancer Inst. 2023. PMID: 36946282 Free PMC article. No abstract available.
References
-
- Shroff RT, Chalasani P, Wei R, et al. Immune responses to COVID-19 mRNA vaccines in patients with solid tumors on active, immunosuppressive cancer therapy. medRxiv. 2021.05.13.21257129. doi: 10.1101/2021.05.13.21257129. - DOI
-
- Deepak P, Kim W, Paley MA, et al. Glucocorticoids and B cell depleting agents substantially impair immunogenicity of mRNA vaccines to SARS-CoV-2. medRxiv. 2021.04.05.21254656. doi: 10.1101/2021.04.05.21254656. - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Miscellaneous

