Microtubule nucleation and γTuRC centrosome localization in interphase cells require ch-TOG
- PMID: 36702836
- PMCID: PMC9879976
- DOI: 10.1038/s41467-023-35955-w
Microtubule nucleation and γTuRC centrosome localization in interphase cells require ch-TOG
Abstract
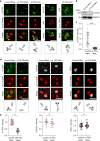
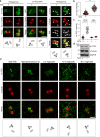
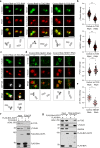
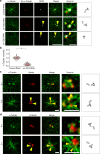
Organization of microtubule arrays requires spatio-temporal regulation of the microtubule nucleator γ-tubulin ring complex (γTuRC) at microtubule organizing centers (MTOCs). MTOC-localized adapter proteins are thought to recruit and activate γTuRC, but the molecular underpinnings remain obscure. Here we show that at interphase centrosomes, rather than adapters, the microtubule polymerase ch-TOG (also named chTOG or CKAP5) ultimately controls γTuRC recruitment and activation. ch-TOG co-assembles with γTuRC to stimulate nucleation around centrioles. In the absence of ch-TOG, γTuRC fails to localize to these sites, but not the centriole lumen. However, whereas some ch-TOG is stably bound at subdistal appendages, it only transiently associates with PCM. ch-TOG's dynamic behavior requires its tubulin-binding TOG domains and a C-terminal region involved in localization. In addition, ch-TOG also promotes nucleation from the Golgi. Thus, at interphase centrosomes stimulation of nucleation and γTuRC attachment are mechanistically coupled through transient recruitment of ch-TOG, and ch-TOG's nucleation-promoting activity is not restricted to centrosomes.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Sub-centrosomal mapping identifies augmin-γTuRC as part of a centriole-stabilizing scaffold.Nat Commun. 2021 Oct 15;12(1):6042. doi: 10.1038/s41467-021-26252-5. Nat Commun. 2021. PMID: 34654813 Free PMC article.
-
The catalytic subunit of DNA polymerase δ inhibits γTuRC activity and regulates Golgi-derived microtubules.Nat Commun. 2017 Sep 15;8(1):554. doi: 10.1038/s41467-017-00694-2. Nat Commun. 2017. PMID: 28916777 Free PMC article.
-
TACC3-ch-TOG interaction regulates spindle microtubule assembly by controlling centrosomal recruitment of γ-TuRC.Biosci Rep. 2023 Mar 29;43(3):BSR20221882. doi: 10.1042/BSR20221882. Biosci Rep. 2023. PMID: 36790370 Free PMC article.
-
Regulation of microtubule nucleation mediated by γ-tubulin complexes.Protoplasma. 2017 May;254(3):1187-1199. doi: 10.1007/s00709-016-1070-z. Epub 2017 Jan 10. Protoplasma. 2017. PMID: 28074286 Review.
-
The augmin connection in the geometry of microtubule networks.Curr Biol. 2015 Mar 30;25(7):R294-9. doi: 10.1016/j.cub.2015.02.006. Curr Biol. 2015. PMID: 25829017 Review.
Cited by
-
Developmental and tissue-specific roles of mammalian centrosomes.FEBS J. 2025 Feb;292(4):709-726. doi: 10.1111/febs.17212. Epub 2024 Jun 27. FEBS J. 2025. PMID: 38935637 Free PMC article. Review.
-
Structural characterization and inhibition of the interaction between ch-TOG and TACC3.J Cell Biol. 2025 Jun 2;224(6):e202407002. doi: 10.1083/jcb.202407002. Epub 2025 Mar 19. J Cell Biol. 2025. PMID: 40105698 Free PMC article.
-
Parthenolide disrupts mitosis by inhibiting ZNF207/BUGZ-promoted kinetochore-microtubule attachment.EMBO J. 2025 Jul;44(13):3764-3793. doi: 10.1038/s44318-025-00469-2. Epub 2025 May 27. EMBO J. 2025. PMID: 40425854 Free PMC article.
-
PLK1 inhibition delays mitotic entry revealing changes to the phosphoproteome of mammalian cells early in division.EMBO J. 2025 Apr;44(7):1891-1920. doi: 10.1038/s44318-025-00400-9. Epub 2025 Mar 3. EMBO J. 2025. PMID: 40033019 Free PMC article.
-
Loss of Eml1 alters microtubule-associated protein networks in mouse brain heterotopia.Commun Biol. 2025 Jul 3;8(1):989. doi: 10.1038/s42003-025-08394-0. Commun Biol. 2025. PMID: 40610677 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

