Microbial dynamics with CRC progression: a study of the mucosal microbiota at multiple sites in cancers, adenomatous polyps, and healthy controls
- PMID: 36703031
- PMCID: PMC9899194
- DOI: 10.1007/s10096-023-04551-7
Microbial dynamics with CRC progression: a study of the mucosal microbiota at multiple sites in cancers, adenomatous polyps, and healthy controls
Abstract
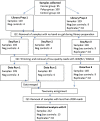
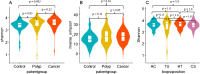
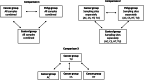
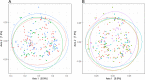
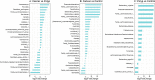
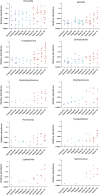
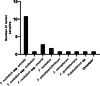
Accumulating evidence has related the gut microbiota to colorectal cancer (CRC). Fusobacterium nucleatum has repeatedly been linked to colorectal tumorigenesis. The aim of this study was to investigate microbial composition in different sampling sites, in order to profile the microbial dynamics with CRC progression. Further, we characterized the tumor-associated F. nucleatum subspecies. Here, we conducted Illumina Miseq next-generation sequencing of the 16S rRNA V4 region in biopsy samples, to investigate microbiota alterations in cancer patients, patients with adenomatous polyp, and healthy controls in Norway. Further, Fusobacterium positive tumor biopsies were subjected to MinION nanopore sequencing of Fusobacterium-specific amplicons to characterize the Fusobacterium species and subspecies. We found enrichment of oral biofilm-associated bacteria, Fusobacterium, Gemella, Parvimonas, Granulicatella, Leptotrichia, Peptostreptococcus, Campylobacter, Selenomonas, Porphyromonas, and Prevotella in cancer patients compared to adenomatous polyp patients and control patients. Higher abundance of amplicon sequence variants (ASVs) classified as Phascolarctobacterium, Bacteroides vulgatus, Bacteroides plebeius, Bacteroides eggerthii, Tyzzerella, Desulfovibrio, Frisingicoccus, Eubacterium coprostanoligenes group, and Lachnospiraceae were identified in cancer and adenomatous polyp patients compared to healthy controls. F. nucleatum ssp. animalis was the dominating subspecies. F. nucleatum ssp. nucleatum, F. nucleatum ssp. vincentii, Fusobacterium pseudoperiodonticum, Fusobacterium necrophorum, and Fusobacterium gonidiaformans were identified in five samples. Several biofilm-associated bacteria were enriched at multiple sites in cancer patients. Another group of bacteria was enriched in both cancer and polyps, suggesting that they may have a role in polyp development and possibly early stages of CRC.
Keywords: 16S rRNA amplicon sequencing; Biofilm bacteria; Colorectal cancer; Fusobacterium species and subspecies; Tumor biopsies.
© 2023. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures


Similar articles
-
Enrichment of Prevotella intermedia in human colorectal cancer and its additive effects with Fusobacterium nucleatum on the malignant transformation of colorectal adenomas.J Biomed Sci. 2022 Oct 27;29(1):88. doi: 10.1186/s12929-022-00869-0. J Biomed Sci. 2022. PMID: 36303164 Free PMC article.
-
Comparative Analysis of Colon Cancer-Derived Fusobacterium nucleatum Subspecies: Inflammation and Colon Tumorigenesis in Murine Models.mBio. 2021 Feb 22;13(1):e0299121. doi: 10.1128/mbio.02991-21. Epub 2022 Feb 8. mBio. 2021. PMID: 35130731 Free PMC article.
-
Fusobacterium Nucleatum Subspecies Animalis Influences Proinflammatory Cytokine Expression and Monocyte Activation in Human Colorectal Tumors.Cancer Prev Res (Phila). 2017 Jul;10(7):398-409. doi: 10.1158/1940-6207.CAPR-16-0178. Epub 2017 May 8. Cancer Prev Res (Phila). 2017. PMID: 28483840
-
Fusobacterium nucleatum, a key pathogenic factor and microbial biomarker for colorectal cancer.Trends Microbiol. 2023 Feb;31(2):159-172. doi: 10.1016/j.tim.2022.08.010. Epub 2022 Sep 1. Trends Microbiol. 2023. PMID: 36058786 Review.
-
The Role of Fusobacterium nucleatum in Colorectal Carcinogenesis.Pathobiology. 2021;88(2):127-140. doi: 10.1159/000512175. Epub 2020 Dec 8. Pathobiology. 2021. PMID: 33291114 Review.
Cited by
-
Intestinal Dysbiosis: Microbial Imbalance Impacts on Colorectal Cancer Initiation, Progression and Disease Mitigation.Biomedicines. 2024 Mar 26;12(4):740. doi: 10.3390/biomedicines12040740. Biomedicines. 2024. PMID: 38672096 Free PMC article. Review.
-
Species-level verification of Phascolarctobacterium association with colorectal cancer.mSystems. 2024 Oct 22;9(10):e0073424. doi: 10.1128/msystems.00734-24. Epub 2024 Sep 17. mSystems. 2024. PMID: 39287376 Free PMC article.
-
Identification of colorectal cancer progression-associated intestinal microbiome and predictive signature construction.J Transl Med. 2023 Jun 8;21(1):373. doi: 10.1186/s12967-023-04119-1. J Transl Med. 2023. PMID: 37291572 Free PMC article.
-
Colorectal cancer biofilm composition reveals distinct bacterial species signature.Appl Microbiol Biotechnol. 2025 Jul 2;109(1):159. doi: 10.1007/s00253-025-13537-8. Appl Microbiol Biotechnol. 2025. PMID: 40603621 Free PMC article.
-
Compositional and functional changes in the salivary microbiota related to oral leukoplakia and oral squamous cell carcinoma: a case control study.BMC Oral Health. 2023 Dec 19;23(1):1021. doi: 10.1186/s12903-023-03760-y. BMC Oral Health. 2023. PMID: 38115005 Free PMC article.
References
-
- Cancer in Norway (2021) Cancer Registry of Norway. Available from: https://www.kreftregisteret.no/en/General/Publications/Cancer-in-Norway/.... Accessed 19 Sept 2022
-
- Flanagan L, Schmid J, Ebert M, Soucek P, Kunicka T, Liska V, et al. Fusobacterium nucleatum associates with stages of colorectal neoplasia development, colorectal cancer and disease outcome. Eur J Clin Microbiol Infect Dis. 2014;33(8):1381–1390. - PubMed
-
- Yang Y, Weng W, Peng J, Hong L, Yang L, Toiyama Y, et al. Fusobacterium nucleatum Increases proliferation of colorectal cancer cells and tumor development in mice by activating toll-like receptor 4 signaling to nuclear factor−κB, and up-regulating expression of microRNA-21. Gastroenterology. 2017;152(4):851–66.e24. - PMC - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical
Molecular Biology Databases

