Impact of engineering the ATP synthase rotor ring on photosynthesis in tobacco chloroplasts
- PMID: 36703219
- PMCID: PMC10231360
- DOI: 10.1093/plphys/kiad043
Impact of engineering the ATP synthase rotor ring on photosynthesis in tobacco chloroplasts
Abstract
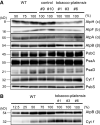
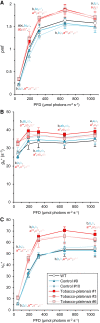
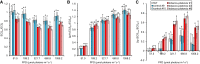
The chloroplast ATP synthase produces the ATP needed for photosynthesis and plant growth. The trans-membrane flow of protons through the ATP synthase rotates an oligomeric assembly of c subunits, the c-ring. The ion-to-ATP ratio in rotary F1F0-ATP synthases is defined by the number of c-subunits in the rotor c-ring. Engineering the c-ring stoichiometry is, therefore, a possible route to manipulate ATP synthesis by the ATP synthase and hence photosynthetic efficiency in plants. Here, we describe the construction of a tobacco (Nicotiana tabacum) chloroplast atpH (chloroplastic ATP synthase subunit c gene) mutant in which the c-ring stoichiometry was increased from 14 to 15 c-subunits. Although the abundance of the ATP synthase was decreased to 25% of wild-type (WT) levels, the mutant lines grew as well as WT plants and photosynthetic electron transport remained unaffected. To synthesize the necessary ATP for growth, we found that the contribution of the membrane potential to the proton motive force was enhanced to ensure a higher proton flux via the c15-ring without unwanted low pH-induced feedback inhibition of electron transport. Our work opens avenues to manipulate plant ion-to-ATP ratios with potentially beneficial consequences for photosynthesis.
© American Society of Plant Biologists 2023. All rights reserved. For permissions, please e-mail: journals.permissions@oup.com.
Conflict of interest statement
Conflict of interest statement. The authors declare no conflicts of interest associated with this manuscript.
Figures


Similar articles
-
Characterization of mutants deficient in N-terminal phosphorylation of the chloroplast ATP synthase subunit β.Plant Physiol. 2023 Mar 17;191(3):1818-1835. doi: 10.1093/plphys/kiad013. Plant Physiol. 2023. PMID: 36635853 Free PMC article.
-
Interaction between PVY HC-Pro and the NtCF1β-subunit reduces the amount of chloroplast ATP synthase in virus-infected tobacco.Sci Rep. 2015 Oct 26;5:15605. doi: 10.1038/srep15605. Sci Rep. 2015. PMID: 26499367 Free PMC article.
-
The roles of ATP synthase and the cytochrome b6/f complexes in limiting chloroplast electron transport and determining photosynthetic capacity.Plant Physiol. 2011 Feb;155(2):956-62. doi: 10.1104/pp.110.168435. Epub 2010 Dec 21. Plant Physiol. 2011. PMID: 21177473 Free PMC article.
-
Structure and Mechanisms of F-Type ATP Synthases.Annu Rev Biochem. 2019 Jun 20;88:515-549. doi: 10.1146/annurev-biochem-013118-110903. Epub 2019 Mar 22. Annu Rev Biochem. 2019. PMID: 30901262 Review.
-
Chloroplast ATP synthase: From structure to engineering.Plant Cell. 2024 Oct 3;36(10):3974-3996. doi: 10.1093/plcell/koae081. Plant Cell. 2024. PMID: 38484126 Free PMC article. Review.
Cited by
-
EngineeRING the gear of ATP synthase: a way to address complexity and flexibility of photosynthesis.Plant Physiol. 2023 May 31;192(2):691-693. doi: 10.1093/plphys/kiad148. Plant Physiol. 2023. PMID: 36880386 Free PMC article. No abstract available.
-
Comparative chloroplast genomes of Argentina species: genome evolution and phylogenomic implications.Front Plant Sci. 2024 Apr 30;15:1349358. doi: 10.3389/fpls.2024.1349358. eCollection 2024. Front Plant Sci. 2024. PMID: 38766467 Free PMC article.
-
Assessing the Impacts of Cu and Mo Engineered Nanomaterials on Crop Plant Growth Using a Targeted Proteomics Approach.ACS Agric Sci Technol. 2023 Dec 22;4(1):103-117. doi: 10.1021/acsagscitech.3c00431. eCollection 2024 Jan 15. ACS Agric Sci Technol. 2023. PMID: 38239573 Free PMC article.
-
Editorial: Functions, working mechanisms, and regulation of rotary ATPases and Ductin proteins.Front Mol Biosci. 2024 Mar 28;11:1399421. doi: 10.3389/fmolb.2024.1399421. eCollection 2024. Front Mol Biosci. 2024. PMID: 38606286 Free PMC article. No abstract available.
-
Transcriptome and anatomical analysis of Stipa breviflora in response to different grazing intensities in desert steppe.Front Plant Sci. 2024 Jun 10;15:1414093. doi: 10.3389/fpls.2024.1414093. eCollection 2024. Front Plant Sci. 2024. PMID: 38916033 Free PMC article.
References
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

