Organophosphorus zwitterions engaged in a conjugated macrocycle on fullerene
- PMID: 36703330
- PMCID: PMC9814461
- DOI: 10.1038/s42004-020-00340-x
Organophosphorus zwitterions engaged in a conjugated macrocycle on fullerene
Abstract
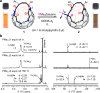
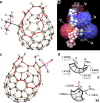
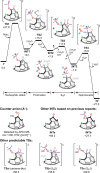
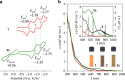
Organophosphorus zwitterions are one of the most important but elusive intermediates for carbon-carbon bond formation in synthetic chemistry and biology. However, a lack of isolated examples due to their lability has hampered in-depth understanding of structures and their reaction mechanisms. In this study, we crystallographically reveal the solid-state structure of a phosha-Michael adduct engaged in a cage-opened C60 skeleton, which is formed as a kinetic product. This compound exhibits dark brown colour in solution with an intense absorption band that extends to 1000 nm, reflecting intramolecular charge transfer transitions. From the 1,2-dicarbonyl moiety on the conjugated orifice, β-oxo-phosphorus ylide is formed as a thermodynamic product. The reaction mechanism that has long been disputed is examined by experimental and theoretical studies, showing a pathway which includes an SN2 reaction as a key step instead of the hitherto considered carbene pathway.
© 2020. The Author(s).
Conflict of interest statement
The authors declare no competing interests.
Figures
Similar articles
-
Nonclassical Abramov Products Formed on Orifices of Cage-Opened C60 Derivatives.Chemistry. 2021 Mar 12;27(15):4864-4868. doi: 10.1002/chem.202004035. Epub 2021 Jan 7. Chemistry. 2021. PMID: 33258190
-
Reactions on a 1,2-Dicarbonyl Moiety of a Fullerene Skeleton.Chemistry. 2021 May 3;27(25):7235-7238. doi: 10.1002/chem.202100640. Epub 2021 Apr 12. Chemistry. 2021. PMID: 33836109
-
Amino-Functionalized Cage-Opened C60 Derivatives.Org Lett. 2021 Dec 17;23(24):9586-9590. doi: 10.1021/acs.orglett.1c03798. Epub 2021 Dec 2. Org Lett. 2021. PMID: 34854690
-
Manganese-Oxygen Intermediates in O-O Bond Activation and Hydrogen-Atom Transfer Reactions.Acc Chem Res. 2017 Nov 21;50(11):2706-2717. doi: 10.1021/acs.accounts.7b00343. Epub 2017 Oct 24. Acc Chem Res. 2017. PMID: 29064667 Review.
-
Fullerene ion chemistry: a journey of discovery and achievement.Philos Trans A Math Phys Eng Sci. 2016 Sep 13;374(2076):20150321. doi: 10.1098/rsta.2015.0321. Philos Trans A Math Phys Eng Sci. 2016. PMID: 27501972 Free PMC article. Review.
Cited by
-
Cation recognition on a fullerene-based macrocycle.Chem Sci. 2020 Oct 22;11(46):12428-12435. doi: 10.1039/d0sc05280a. Chem Sci. 2020. PMID: 34123228 Free PMC article.
-
Synthesis of inter-[60]fullerene conjugates with inherent chirality.Nat Commun. 2024 Jan 15;15(1):514. doi: 10.1038/s41467-024-44834-x. Nat Commun. 2024. PMID: 38225251 Free PMC article.
-
Open-[60]fullerene-aniline conjugates with near-infrared absorption.RSC Adv. 2023 May 12;13(21):14575-14579. doi: 10.1039/d3ra02113k. eCollection 2023 May 9. RSC Adv. 2023. PMID: 37188248 Free PMC article.
-
An orifice design: water insertion into C60.RSC Adv. 2020 Nov 6;10(66):40406-40410. doi: 10.1039/d0ra09067k. eCollection 2020 Nov 2. RSC Adv. 2020. PMID: 35520847 Free PMC article.
References
-
- Wittig G, Geisslerg G. Course of reactions of pentaphenylphosphorus and certain derivatives. Justus Liebigs Ann. Chem. 1953;580:44–57. doi: 10.1002/jlac.19535800107. - DOI
LinkOut - more resources
Full Text Sources

