Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex
- PMID: 36703411
- PMCID: PMC9814094
- DOI: 10.1038/s42004-020-0287-0
Iodide-enhanced palladium catalysis via formation of iodide-bridged binuclear palladium complex
Abstract
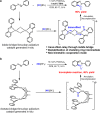
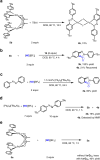
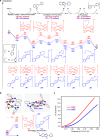
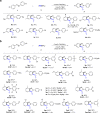
The prevalence of metalloenzymes with multinuclear metal complexes in their active sites inspires chemists' interest in the development of multinuclear catalysts. Studies in this area commonly focus on binuclear catalysts containing either metal-metal bond or electronically discrete, conformationally advantageous metal centres connected by multidentate ligands, while in many multinuclear metalloenzymes the metal centres are bridged through μ2-ligands without a metal-metal bond. We report herein a μ2-iodide-bridged binuclear palladium catalyst which accelerates the C-H nitrosation/annulation reaction and significantly enhances its yield compared with palladium acetate catalyst. The superior activity of this binuclear palladium catalyst is attributed to the trans effect-relay through the iodide bridge from one palladium sphere to the other palladium sphere, which facilitates dissociation of the stable six-membered chelating ring in palladium intermediate and accelerates the catalytic cycle. Such a trans effect-relay represents a bimetallic cooperation mode and may open an avenue to design and develop multinuclear catalysts.
© 2020. The Author(s).
Conflict of interest statement
The authors declare no competing interests
Figures

Similar articles
-
Bimetallic redox synergy in oxidative palladium catalysis.Acc Chem Res. 2012 Jun 19;45(6):840-50. doi: 10.1021/ar2001974. Epub 2011 Oct 27. Acc Chem Res. 2012. PMID: 22029861
-
Recent advances in sustainable N-heterocyclic carbene-Pd(II)-pyridine (PEPPSI) catalysts: A review.Environ Res. 2023 May 15;225:115515. doi: 10.1016/j.envres.2023.115515. Epub 2023 Feb 24. Environ Res. 2023. PMID: 36842701 Review.
-
Pentanuclear Scaffold: A Molecular Platform for Small-Molecule Conversions.Acc Chem Res. 2020 Oct 20;53(10):2140-2151. doi: 10.1021/acs.accounts.0c00186. Epub 2020 Sep 1. Acc Chem Res. 2020. PMID: 32870647
-
Transition metal chemistry of cyclodiphosphanes containing phosphine and amide-phosphine functionalities: formation of a stable dipalladium(II) complex containing a Pd-P σ-bond.Dalton Trans. 2010 Dec 14;39(46):11149-62. doi: 10.1039/c0dt00614a. Epub 2010 Oct 8. Dalton Trans. 2010. PMID: 20936211
-
Dinuclear Nickel(I) and Palladium(I) Complexes for Highly Active Transformations of Organic Compounds.Molecules. 2018 Jan 11;23(1):140. doi: 10.3390/molecules23010140. Molecules. 2018. PMID: 29324677 Free PMC article. Review.
References
-
- Shibasaki M, Sasai H, Arai T. Asymmetric catalysis with heterobimetallic compounds. Angew. Chem. Int. Ed. 1997;36:1236–1256. doi: 10.1002/anie.199712361. - DOI
-
- van den Beuken EK, Feringa BL. Bimetallic catalysis by late transition metal complexes. Tetrahedron. 1998;54:12985–13011. doi: 10.1016/S0040-4020(98)00319-6. - DOI
LinkOut - more resources
Full Text Sources

