Brief research report: Repurposing pentoxifylline to treat intense acute swimming-Induced delayed-onset muscle soreness in mice: Targeting peripheral and spinal cord nociceptive mechanisms
- PMID: 36703752
- PMCID: PMC9871252
- DOI: 10.3389/fphar.2022.950314
Brief research report: Repurposing pentoxifylline to treat intense acute swimming-Induced delayed-onset muscle soreness in mice: Targeting peripheral and spinal cord nociceptive mechanisms
Abstract
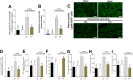
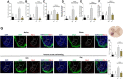
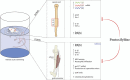
In this study, we pursue determining the effect of pentoxifylline (Ptx) in delayed-onset muscle soreness (DOMS) triggered by exposing untrained mice to intense acute swimming exercise (120 min), which, to our knowledge, has not been investigated. Ptx treatment (1.5, 4.5, and 13.5 mg/kg; i.p., 30 min before and 12 h after the session) reduced intense acute swimming-induced mechanical hyperalgesia in a dose-dependent manner. The selected dose of Ptx (4.5 mg/kg) inhibited recruitment of neutrophils to the muscle tissue, oxidative stress, and both pro- and anti-inflammatory cytokine production in the soleus muscle and spinal cord. Furthermore, Ptx treatment also reduced spinal cord glial cell activation. In conclusion, Ptx reduces pain by targeting peripheral and spinal cord mechanisms of DOMS.
Keywords: cytokine; glial cells; muscle mechanical hyperalgesia; oxidative stress; pentoxifylline.
Copyright © 2023 Borghi, Zaninelli, Saraiva-Santos, Bertozzi, Cardoso, Carvalho, Ferraz, Camilios-Neto, Cunha, Cunha, Pinho-Ribeiro, Casagrande and Verri.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Targeting interleukin-1β reduces intense acute swimming-induced muscle mechanical hyperalgesia in mice.J Pharm Pharmacol. 2014 Jul;66(7):1009-20. doi: 10.1111/jphp.12226. Epub 2014 Feb 13. J Pharm Pharmacol. 2014. PMID: 24697255
-
Role of TNF-α/TNFR1 in intense acute swimming-induced delayed onset muscle soreness in mice.Physiol Behav. 2014 Apr 10;128:277-87. doi: 10.1016/j.physbeh.2014.01.023. Epub 2014 Feb 8. Physiol Behav. 2014. PMID: 24518865
-
Intense Acute Swimming Induces Delayed-Onset Muscle Soreness Dependent on Spinal Cord Neuroinflammation.Front Pharmacol. 2022 Jan 7;12:734091. doi: 10.3389/fphar.2021.734091. eCollection 2021. Front Pharmacol. 2022. PMID: 35069187 Free PMC article.
-
Delayed onset muscle soreness : treatment strategies and performance factors.Sports Med. 2003;33(2):145-64. doi: 10.2165/00007256-200333020-00005. Sports Med. 2003. PMID: 12617692 Review.
-
Does Acupuncture Benefit Delayed-Onset Muscle Soreness After Strenuous Exercise? A Systematic Review and Meta-Analysis.Front Physiol. 2020 Jul 17;11:666. doi: 10.3389/fphys.2020.00666. eCollection 2020. Front Physiol. 2020. PMID: 32765287 Free PMC article.
Cited by
-
The Role of Exercise on Glial Cell Activity in Neuropathic Pain Management.Cells. 2025 Mar 24;14(7):487. doi: 10.3390/cells14070487. Cells. 2025. PMID: 40214441 Free PMC article. Review.
-
Role of TRPV1+ and TRPA1+ nociceptive neurons in delayed-onset muscle soreness: inhibition by hesperidin methyl chalcone.Inflammopharmacology. 2025 May;33(5):2815-2832. doi: 10.1007/s10787-025-01762-6. Epub 2025 Apr 26. Inflammopharmacology. 2025. PMID: 40285987
References
LinkOut - more resources
Full Text Sources
Medical

