Oral pretreatment with β-lactoglobulin derived peptide and CpG co-encapsulated in PLGA nanoparticles prior to sensitizations attenuates cow's milk allergy development in mice
- PMID: 36703973
- PMCID: PMC9872660
- DOI: 10.3389/fimmu.2022.1053107
Oral pretreatment with β-lactoglobulin derived peptide and CpG co-encapsulated in PLGA nanoparticles prior to sensitizations attenuates cow's milk allergy development in mice
Abstract
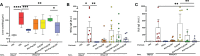
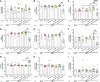
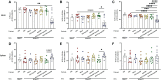
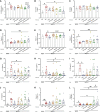
Cow's milk allergy is a common food allergy among infants. Improved hygiene conditions and loss of microbial diversity are associated with increased risk of allergy development. The intestinal immune system is essential for oral tolerance induction. In this respect, bacterial CpG DNA is known to drive Th1 and regulatory T-cell (Treg) development via Toll-Like-Receptor 9 (TLR-9) signaling, skewing away from the allergic Th2 phenotype. We aimed to induce allergen specific tolerance via oral delivery of poly (lactic-co-glycolic acid) nanoparticles (NP) co-encapsulated with a selected β-lactoglobulin derived peptide (BLG-Pep) and TLR-9 ligand CpG oligodeoxynucleotide (CpG). In vivo, 3-4-week-old female C3H/HeOuJ mice housed in individually ventilated cages received 6-consecutive-daily gavages of either PBS, whey, BLG-Pep/NP, CpG/NP, a mixture of BLG-Pep/NP plus CpG/NP or co-encapsulated BLG-Pep+CpG/NP, before 5-weekly oral sensitizations with whey plus cholera toxin (CT) or only CT (sham) and were challenged with whey 5 days after the last sensitization. The co-encapsulated BLG-Pep+CpG/NP pretreatment, but not BLG-Pep/NP, CpG/NP or the mixture of BLG-Pep/NP plus CpG/NP, prevented the whey-induced allergic skin reactivity and prevented rise in serum BLG-specific IgE compared to whey-sensitized mice. Importantly, co-encapsulated BLG-Pep+CpG/NP pretreatment reduced dendritic cell (DC) activation and lowered the frequencies of PD-L1+ DC in the mesenteric lymph nodes compared to whey-sensitized mice. By contrast, co-encapsulated BLG-Pep+CpG/NP pretreatment increased the frequency of splenic PD-L1+ DC compared to the BLG-Pep/NP plus CpG/NP recipients, in association with lower Th2 development and increased Treg/Th2 and Th1/Th2 ratios in the spleen. Oral administration of PLGA NP co-encapsulated with BLG-Pep and CpG prevented rise in serum BLG-specific IgE and symptom development while lowering splenic Th2 cell frequency in these mice which were kept under strict hygienic conditions.
Keywords: CpG oligodeoxynucleotides; cow’s milk allergy; individual ventilated cages; oral pretreatment; poly (lactic-co-glycolic acid) nanoparticles; toll-like-receptor 9; whey protein; β-lactoglobulin.
Copyright © 2023 Liu, Thijssen, Hennink, Garssen, van Nostrum and Willemsen.
Conflict of interest statement
JG is employed by Nutricia Research B.V. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


Similar articles
-
PLGA nanoparticles loaded with beta-lactoglobulin-derived peptides modulate mucosal immunity and may facilitate cow's milk allergy prevention.Eur J Pharmacol. 2018 Jan 5;818:211-220. doi: 10.1016/j.ejphar.2017.10.051. Epub 2017 Oct 25. Eur J Pharmacol. 2018. PMID: 29079360
-
Inhibition of cow's milk allergy development in mice by oral delivery of β-lactoglobulin-derived peptides loaded PLGA nanoparticles is associated with systemic whey-specific immune silencing.Clin Exp Allergy. 2022 Jan;52(1):137-148. doi: 10.1111/cea.13967. Epub 2021 Jul 1. Clin Exp Allergy. 2022. PMID: 34145667 Free PMC article.
-
A Specific Mixture of Fructo-Oligosaccharides and Bifidobacterium breve M-16V Facilitates Partial Non-Responsiveness to Whey Protein in Mice Orally Exposed to β-Lactoglobulin-Derived Peptides.Front Immunol. 2017 Jan 12;7:673. doi: 10.3389/fimmu.2016.00673. eCollection 2016. Front Immunol. 2017. PMID: 28127297 Free PMC article.
-
Recent perspective on cow's milk allergy and dairy nutrition.Crit Rev Food Sci Nutr. 2022;62(27):7503-7517. doi: 10.1080/10408398.2021.1915241. Epub 2021 May 13. Crit Rev Food Sci Nutr. 2022. PMID: 33983082 Review.
-
Research Progress of Oral Immune Tolerance Mechanism Induced by Whey Protein.Nutrients. 2025 Apr 29;17(9):1517. doi: 10.3390/nu17091517. Nutrients. 2025. PMID: 40362825 Free PMC article. Review.
Cited by
-
Exopolysaccharide β-(2,6)-levan-type fructans have a molecular-weight-dependent modulatory effect on Toll-like receptor signalling.Food Funct. 2024 Jan 22;15(2):676-688. doi: 10.1039/d3fo03066k. Food Funct. 2024. PMID: 38108152 Free PMC article.
-
Live Cell Imaging by Förster Resonance Energy Transfer Fluorescence to Study Trafficking of PLGA Nanoparticles and the Release of a Loaded Peptide in Dendritic Cells.Pharmaceuticals (Basel). 2023 May 31;16(6):818. doi: 10.3390/ph16060818. Pharmaceuticals (Basel). 2023. PMID: 37375766 Free PMC article.
-
Modulation of immune response by nanoparticle-based immunotherapy against food allergens.Front Immunol. 2023 Sep 8;14:1229667. doi: 10.3389/fimmu.2023.1229667. eCollection 2023. Front Immunol. 2023. PMID: 37744376 Free PMC article. Review.
-
Scientific developments in understanding food allergy prevention, diagnosis, and treatment.Front Immunol. 2025 Apr 22;16:1572283. doi: 10.3389/fimmu.2025.1572283. eCollection 2025. Front Immunol. 2025. PMID: 40330465 Free PMC article. Review.
-
Advances in dendritic cell targeting nano-delivery systems for induction of immune tolerance.Front Bioeng Biotechnol. 2023 Oct 9;11:1242126. doi: 10.3389/fbioe.2023.1242126. eCollection 2023. Front Bioeng Biotechnol. 2023. PMID: 37877041 Free PMC article. Review.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

