Evaluation of the osteogenic effect of apigenin on human mesenchymal stem cells by inhibiting inflammation through modulation of NF-κB/IκBα
- PMID: 36704428
- PMCID: PMC9872176
- DOI: 10.4103/1735-5362.359436
Evaluation of the osteogenic effect of apigenin on human mesenchymal stem cells by inhibiting inflammation through modulation of NF-κB/IκBα
Abstract
Background and purpose: Apigenin has stimulatory effects on osteogenic differentiation of human mesenchymal stem cells (hMSCs) as well as anti-inflammatory properties. This study investigated the osteogenic differentiation of hMSCs in inflammatory conditions treated with apigenin focusing on nuclear factor kappa-light-chain-enhancer of activated B (NF-кB), nuclear factor of kappa light polypeptide gene enhancer in B-cells inhibitor, alpha (IκBα) and nucleotide-binding oligomerization domain, leucine-rich repeat and pyrin domain containing 3 (NLRP3) inflammatory pathways.
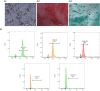
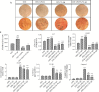
Experimental approach: Along with osteogenic differentiation of the hMSCs, they became inflamed with lipopolysaccharide (LPS)/palmitic acid (PA) and treated with apigenin. Alizarin red staining, alkaline phosphatase (ALP) activity, and Runt-related transcription factor 2 (RUNX2) gene expression were used to determine the degree of differentiation. Also, gene expression of NLRP3 was performed along with protein expression of interleukin 1-beta (IL-1β), NF-кB, and IκBα.
Findings / results: Apigenin was shown to be effective in neutralizing the inhibitory impact of LPS/PA on osteogenesis. Apigenin increased MSC osteogenic capacity by inhibiting NLRP3 expression and the activity of caspase-1. It was also associated with a considerable decrease in the protein expression of NF-κB and IκBα, as well as IL-1β, in these cells.
Conclusion and implications: The effects of apigenin on osteogenesis under inflammatory conditions were cautiously observed.
Keywords: Apigenin; Inflammation; Mesenchymal stem cells; NF-кB; NLRP3; RUNX2.
Copyright: © 2022 Research in Pharmaceutical Sciences.
Figures
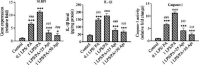
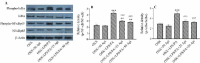
References
LinkOut - more resources
Full Text Sources
